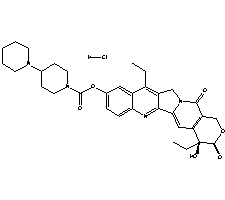
Irinotecan hydrochloride cas no:100286-90-6
Synonyms: [1,4'-Bipiperidine]-1'-carboxylicacid,(4S)-4,11-diethyl-3,4,12,14-tetrahydro-4-hydroxy-3,14-dioxo-1H-pyrano[3',4':6,7]indolizino[1,2-b]quinolin-9-yl ester, monohydrochloride(9CI);[1,4'-Bipiperidine]-1'-carboxylic acid,4,11-diethyl-3,4,12,14-tetrahydro-4-hydroxy-3,14-dioxo-1H-pyrano[3',4':6,7]indolizino[1,2-b]quinolin-9-yl ester, monohydrochloride,(S)-;1H-Pyrano[3',4':6,7]indolizino[1,2-b]quinoline, [1,4'-bipiperidine]-1'-carboxylic acid deriv.;7-Ethyl-10-[[4-(1-piperidyl)-1-piperidyl]carbonyloxy]camptothecin hydrochloride;CPT 11;Campto;Camptosar;Camptothecin 11;Camptothecin 11 hydrochloride;Topotecin;U 101440E;
NameIrinotecan hydrochloride
CAS100286-90-6
Synonyms[1,4'-Bipiperidine]-1'-carboxylicacid,(4S)-4,11-diethyl-3,4,12,14-tetrahydro-4-hydroxy-3,14-dioxo-1H-pyrano[3',4':6,7]indolizino[1,2-b]quinolin-9-yl ester, monohydrochloride(9CI);[1,4'-Bipiperidine]-1'-carboxylic acid,4,11-diethyl-3,4,12,14-tetrahydro-4-hydroxy-3,14-dioxo-1H-pyrano[3',4':6,7]indolizino[1,2-b]quinolin-9-yl ester, monohydrochloride,(S)-;1H-Pyrano[3',4':6,7]indolizino[1,2-b]quinoline, [1,4'-bipiperidine]-1'-carboxylic acid deriv.;7-Ethyl-10-[[4-(1-piperidyl)-1-piperidyl]carbonyloxy]camptothecin hydrochloride;CPT 11;Campto;Camptosar;Camptothecin 11;Camptothecin 11 hydrochloride;Topotecin;U 101440E;
Molecular FormulaC33H38N4O6.HCl
Molecular Weight623.15
AppearanceYellow crystalline powder
refractive index67.7 ° (C=1, H2O)
storage tempRefrigerator
CHEMICAL IDENTIFICATION
- RTECS NUMBER :
- DW1061000
- CAS REGISTRY NUMBER :
- 100286-90-6
- LAST UPDATED :
- 199806
- DATA ITEMS CITED :
- 20
- MOLECULAR FORMULA :
- C33-H38-N4-O6.Cl-H
- MOLECULAR WEIGHT :
- 623.21
HEALTH HAZARD DATA
ACUTE TOXICITY DATA
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 867 mg/kg
- TOXIC EFFECTS :
- Sense Organs and Special Senses (Eye) - effect, not otherwise specified
Behavioral - convulsions or effect on seizure threshold
Lungs, Thorax, or Respiration - other changes
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 83600 ug/kg
- TOXIC EFFECTS :
- Tumorigenic - active as anti-cancer agent
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 765 mg/kg
- TOXIC EFFECTS :
- Tumorigenic - active as anti-cancer agent
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intraperitoneal
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 177 mg/kg
- TOXIC EFFECTS :
- Tumorigenic - active as anti-cancer agent
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 132 mg/kg
- TOXIC EFFECTS :
- Sense Organs and Special Senses (Eye) - effect, not otherwise specified
Behavioral - convulsions or effect on seizure threshold
Lungs, Thorax, or Respiration - other changes
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Mammal - dog
- DOSE/DURATION :
- 40 mg/kg
- TOXIC EFFECTS :
- Behavioral - convulsions or effect on seizure threshold
Gastrointestinal - hypermotility, diarrhea
Gastrointestinal - nausea or vomiting
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 728 mg/kg/26W-C
- TOXIC EFFECTS :
- Endocrine - other changes
Blood - changes in serum composition (e.g. TP, bilirubin, cholesterol)
Blood - changes in other cell count (unspecified)
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 112 mg/kg/4W-I
- TOXIC EFFECTS :
- Endocrine - changes in thymus weight
Blood - pigmented or nucleated red blood cells
Blood - other changes
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intraperitoneal
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 700 mg/kg/14D-I
- TOXIC EFFECTS :
- Gastrointestinal - ulceration or bleeding from small intestine
Gastrointestinal - hypermotility, diarrhea
Gastrointestinal - other changes
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Mammal - dog
- DOSE/DURATION :
- 44800 ug/kg/28D-C
- TOXIC EFFECTS :
- Endocrine - changes in spleen weight
Blood - changes in serum composition (e.g. TP, bilirubin, cholesterol)
Blood - changes in other cell count (unspecified)
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravenous
- DOSE :
- 66 mg/kg
- SEX/DURATION :
- female 7-17 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death,
e.g., stunted fetus)
Reproductive - Effects on Embryo or Fetus - fetal death
Reproductive - Specific Developmental Abnormalities - Central Nervous System
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravenous
- DOSE :
- 66 mg/kg
- SEX/DURATION :
- female 7-17 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - Central Nervous System
Reproductive - Specific Developmental Abnormalities - eye/ear
Reproductive - Specific Developmental Abnormalities - craniofacial
(including nose and tongue)
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravenous
- DOSE :
- 66 mg/kg
- SEX/DURATION :
- female 7-17 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - skin and skin
appendages
Reproductive - Specific Developmental Abnormalities - musculoskeletal system
Reproductive - Specific Developmental Abnormalities - cardiovascular
(circulatory) system
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravenous
- DOSE :
- 66 mg/kg
- SEX/DURATION :
- female 7-17 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Newborn - live birth index (measured after birth)
Reproductive - Effects on Newborn - viability index (e.g., # alive at day 4
per # born alive)
Reproductive - Effects on Newborn - growth statistics (e.g.%, reduced weight
gain)
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravenous
- DOSE :
- 78 mg/kg
- SEX/DURATION :
- female 6-18 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetal death
Reproductive - Specific Developmental Abnormalities - craniofacial
(including nose and tongue)
Reproductive - Specific Developmental Abnormalities - cardiovascular
(circulatory) system
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravenous
- DOSE :
- 78 mg/kg
- SEX/DURATION :
- female 6-18 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - musculoskeletal system
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravenous
- DOSE :
- 78 mg/kg
- SEX/DURATION :
- female 6-18 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetal death
Reproductive - Specific Developmental Abnormalities - craniofacial
(including nose and tongue)
Reproductive - Specific Developmental Abnormalities - musculoskeletal system
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravenous
- DOSE :
- 78 mg/kg
- SEX/DURATION :
- female 6-18 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - cardiovascular
(circulatory) system
MUTATION DATA
- TYPE OF TEST :
- DNA inhibition
- TEST SYSTEM :
- Rodent - mouse Leukocyte
- DOSE/DURATION :
- 100 umol/L
- REFERENCE :
- CNREA8 Cancer Research. (Public Ledger Building, Suit 816, 6th & Chestnut
Sts., Philadelphia, PA 19106) V.1- 1941- Volume(issue)/page/year:
51,4187,1991