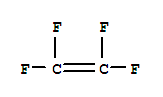Ethene,1,1,2,2-tetrafluoro- cas no:116-14-3
Synonyms: Ethene,tetrafluoro- (9CI);Ethylene, tetrafluoro- (6CI,8CI);1,1,2,2-Tetrafluoroethylene;Ethylene tetrafluoride;Lubron 5;PFC 1114;Perfluoroethene;Perfluoroethylene;R 1114;TFE;Teflon 851K02730;Tetrafluoroethene;Tetrafluoroethylene;
NameEthene,1,1,2,2-tetrafluoro-
CAS116-14-3
SynonymsEthene,tetrafluoro- (9CI);Ethylene, tetrafluoro- (6CI,8CI);1,1,2,2-Tetrafluoroethylene;Ethylene tetrafluoride;Lubron 5;PFC 1114;Perfluoroethene;Perfluoroethylene;R 1114;TFE;Teflon 851K02730;Tetrafluoroethene;Tetrafluoroethylene;
EINECS(EC#)204-126-9
Molecular FormulaC2F4
Molecular Weight100.02
Appearancecolourless gas
refractive index1.241
storage tempCylinders containing tetrafluoroethylene under high pressure.Should be stored in a cool, well-ventilated area and protected against accidental impacts store in a cool, dry, well-ventilated location. Separate from oxidizing materials, air, sulfur trioxide, halogen compounds. Outside or detached storage is preferred.
Globally Harmonized System of Classification adn Labelling of Chemicals(GHS)
Hazardclass2.1
Hazard Flammable, dangerous fire risk.
Risk R11
Safety Confirmed carcinogen. Mildly toxic by inhalation. Can act as an asphyxiant and may have other toxic properties. The gas is flammable when exposed to heat or flame. The inhibited monomer will explode if ignited. Explosive in the form of vapor when exposed to heat or flame. Will explode at pressures above 2.7 bar if limonene inhibitor is not added. Iodine pentafluoride depletes the limonene inhibitor and then causes explosive polymerization of the monomer. Mixtures with hexafluoropropene and air form an explosive peroxide. Reacts violently with SO3; air; difluoromethylene dihypofluorite; dioxygen difluoride; iodine pentafluoride; oxygen. When heated to decomposition it emits highly toxic fumes of F−. See also FLUORIDES.
Inhalation causes irritation of respiratory system. Contact with eyes causes slight irritation.