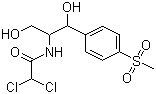
Acetamide,2,2-dichloro-N-[(1R,2R)-2-hydroxy-1-(hydroxymethyl)-2-[4-(methylsulfonyl)phenyl]ethyl]- cas no:15318-45-3
Synonyms: Acetamide,2,2-dichloro-N-[2-hydroxy-1-(hydroxymethyl)-2-[4-(methylsulfonyl)phenyl]ethyl]-,[R-(R*,R*)]-;(Methylsulfonyl)chloramphenicol;8065CB;D-Thiocymetin;D-Thiophenicol;D-d-threo-2-Dichloroacetamido-1-(4-methylsulfonylphenyl)-1,3-propanediol;Dextrosulphenidol;NSC 522822;Thiocymetin;Thiophenicol;Win 5063-2;
NameAcetamide,2,2-dichloro-N-[(1R,2R)-2-hydroxy-1-(hydroxymethyl)-2-[4-(methylsulfonyl)phenyl]ethyl]-
CAS15318-45-3
SynonymsAcetamide,2,2-dichloro-N-[2-hydroxy-1-(hydroxymethyl)-2-[4-(methylsulfonyl)phenyl]ethyl]-,[R-(R*,R*)]-;(Methylsulfonyl)chloramphenicol;8065CB;D-Thiocymetin;D-Thiophenicol;D-d-threo-2-Dichloroacetamido-1-(4-methylsulfonylphenyl)-1,3-propanediol;Dextrosulphenidol;NSC 522822;Thiocymetin;Thiophenicol;Win 5063-2;
EINECS(EC#)239-355-3
Molecular FormulaC12H15Cl2NO5S
Molecular Weight356.22
Appearanceoff-white solid
refractive index1.589
Safety Poison by intravenous route. Moderately toxic by subcutaneous route. Mildly toxic by ingestion. Human systemic effects by an unspecified route: sleep disorders, dermatitis, nausea or vomiting. An experimental teratogen. Experimental reproductive effects. When heated to decomposition it emits very toxic fumes of SOx, NOx, and Cl?. See also CHLORAMPHENICOL.
CHEMICAL IDENTIFICATION
- RTECS NUMBER :
- AB6680000
- CHEMICAL NAME :
- Acetamide,
2,2-dichloro-N-(beta-hydroxy-alpha-(hydroxymethyl)-p-
(methylsulfonyl)phene thyl)-, D-threo-(+)
- CAS REGISTRY NUMBER :
- 15318-45-3
- LAST UPDATED :
- 199806
- DATA ITEMS CITED :
- 16
- MOLECULAR FORMULA :
- C12-H15-Cl2-N-O5-S
- MOLECULAR WEIGHT :
- 356.24
- WISWESSER LINE NOTATION :
- WS1&R DYQY1QMVYGG
HEALTH HAZARD DATA
ACUTE TOXICITY DATA
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Unreported
- SPECIES OBSERVED :
- Human
- DOSE/DURATION :
- 214 mg/kg/10D
- TOXIC EFFECTS :
- Behavioral - sleep
Gastrointestinal - nausea or vomiting
Skin and Appendages - dermatitis, other (after systemic exposure)
- REFERENCE :
- ARZNAD Arzneimittel-Forschung. Drug Research. (Editio Cantor Verlag,
Postfach 1255, W-7960 Aulendorf, Fed. Rep. Ger.) V.1- 1951-
Volume(issue)/page/year: 24,944,1974
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- >5 gm/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- NIIRDN Drugs in Japan (Ethical Drugs). (Yakugyo Jiho Co., Ltd., Tokyo,
Japan) Volume(issue)/page/year: -,740,1995
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intraperitoneal
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- >5 gm/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 3,390,1969
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 339 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 3,390,1969
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- >7 gm/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 3,390,1969
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intraperitoneal
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- >5 gm/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 3,390,1969
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 368 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 3,390,1969
** OTHER MULTIPLE DOSE TOXICITY DATA **
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 3724 mg/kg/4W-C
- TOXIC EFFECTS :
- Gastrointestinal - alteration in gastric secretion
Blood - hemorrhage
Nutritional and Gross Metabolic - weight loss or decreased weight gain
- REFERENCE :
- TOLED5 Toxicology Letters. (Elsevier Science Pub. B.V., POB 211, 1000 AE
Amsterdam, Netherlands) V.1- 1977- Volume(issue)/page/year: 91,137,1997
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 1583 mg/kg/13W-C
- TOXIC EFFECTS :
- Liver - changes in liver weight
Blood - pigmented or nucleated red blood cells
Blood - changes in erythrocyte (RBC) count
- REFERENCE :
- TOLED5 Toxicology Letters. (Elsevier Science Pub. B.V., POB 211, 1000 AE
Amsterdam, Netherlands) V.1- 1977- Volume(issue)/page/year: 91,137,1997
** REPRODUCTIVE DATA **
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 300 mg/kg
- SEX/DURATION :
- female 9-14 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Fertility - post-implantation mortality (e.g. dead and/or
resorbed implants per total number of implants)
Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death,
e.g., stunted fetus)
Reproductive - Effects on Embryo or Fetus - fetal death
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 7,41,1973
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 150 mg/kg
- SEX/DURATION :
- female 9-14 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - musculoskeletal system
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 7,41,1973
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intraperitoneal
- DOSE :
- 400 mg/kg
- SEX/DURATION :
- female 1-7 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Fertility - post-implantation mortality (e.g. dead and/or
resorbed implants per total number of implants)
Reproductive - Fertility - litter size (e.g. # fetuses per litter; measured
before birth)
- REFERENCE :
- FESTAS Fertility and Sterility. (American Fertility Soc., 608 13th Ave. S,
Birmingham, AL 35282) V.1- 1950- Volume(issue)/page/year: 21,431,1970
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- DOSE :
- 400 mg/kg
- SEX/DURATION :
- female 10-11 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - other effects to embryo
- REFERENCE :
- TJADAB Teratology, The International Journal of Abnormal Development. (Alan
R. Liss, Inc., 41 E. 11th St., New York, NY 10003) V.1- 1968-
Volume(issue)/page/year: 18,93,1978
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Unreported
- DOSE :
- 250 mg/kg
- SEX/DURATION :
- female 10-11 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetal death
- REFERENCE :
- NSAPCC Naunyn-Schmiedeberg's Archives of Pharmacology. (Springer Verlag,
Heidelberger, Pl. 3, D-1000 Berlin 33, Fed. Rep. Ger.) V.272- 1972-
Volume(issue)/page/year: 293(Suppl),R60,1976
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 2400 mg/kg
- SEX/DURATION :
- female 7-12 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death,
e.g., stunted fetus)
Reproductive - Specific Developmental Abnormalities - musculoskeletal system
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 7,41,1973
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 6 gm/kg
- SEX/DURATION :
- female 7-12 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Fertility - post-implantation mortality (e.g. dead and/or
resorbed implants per total number of implants)
Reproductive - Effects on Embryo or Fetus - fetal death
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 7,41,1973