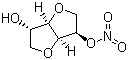
1,4:3,6-dianhydro-D-glucitol 5-nitrate cas no:16051-77-7
Synonyms: Isosorbide mononitrate
Name1,4:3,6-dianhydro-D-glucitol 5-nitrate
CAS16051-77-7
SynonymsIsosorbide mononitrate
EINECS(EC#)240-197-2
Molecular FormulaC6H9NO6
Molecular Weight191.14
Appearancewhite crystalline powder
alpha170 o (C=1, ETOH)
refractive index145 ° (C=5, H2O)
storage tempKeep away from heat, sparks, and flame. Keep away from sources of ignition. Store in a tightly closed container. Store in a cool, dry, well-ventilated area away from incompatible substances.
Globally Harmonized System of Classification adn Labelling of Chemicals(GHS)
Hazardclass1.1A
Hazard F:Flammable
Risk R11;R36/37/38
CHEMICAL IDENTIFICATION
- RTECS NUMBER :
- LZ4386500
- CAS REGISTRY NUMBER :
- 16051-77-7
- LAST UPDATED :
- 199706
- DATA ITEMS CITED :
- 13
- MOLECULAR FORMULA :
- C6-H9-N-O6
- MOLECULAR WEIGHT :
- 191.16
HEALTH HAZARD DATA
ACUTE TOXICITY DATA
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 2010 mg/kg
- TOXIC EFFECTS :
- Behavioral - ataxia
Kidney, Ureter, Bladder - urine volume increased
Nutritional and Gross Metabolic - body temperature decrease
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 29,327,1985
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intraperitoneal
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 1760 mg/kg
- TOXIC EFFECTS :
- Sense Organs and Special Senses (Eye) - lacrimation
Behavioral - altered sleep time (including change in righting reflex)
Nutritional and Gross Metabolic - body temperature decrease
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 29,327,1985
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 1750 mg/kg
- TOXIC EFFECTS :
- Behavioral - altered sleep time (including change in righting reflex)
Behavioral - ataxia
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 29,327,1985
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 1771 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- BCFAAI Bollettino Chimico Farmaceutico. (Societa Editoriale Farmaceutica,
Via Ausonio 12, 20123 Milan, Italy) V.33- 1894- Volume(issue)/page/year:
128,74,1989
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intraperitoneal
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 1810 mg/kg
- TOXIC EFFECTS :
- Behavioral - ataxia
Skin and Appendages - hair
Nutritional and Gross Metabolic - weight loss or decreased weight gain
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 29,327,1985
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 1820 mg/kg
- TOXIC EFFECTS :
- Behavioral - altered sleep time (including change in righting reflex)
Behavioral - ataxia
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 29,327,1985
** OTHER MULTIPLE DOSE TOXICITY DATA **
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 147 gm/kg/26W-C
- TOXIC EFFECTS :
- Kidney, Ureter, Bladder - other changes in urine composition
Endocrine - changes in spleen weight
Blood - other changes
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 33,171,1987
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 182 gm/kg/13W-C
- TOXIC EFFECTS :
- Liver - changes in liver weight
Blood - changes in serum composition (e.g. TP, bilirubin, cholesterol)
Blood - other changes
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 30,1079,1985
** REPRODUCTIVE DATA **
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 91 gm/kg
- SEX/DURATION :
- male 91 day(s) pre-mating
- TOXIC EFFECTS :
- Reproductive - Paternal Effects - testes, epididymis, sperm duct
Reproductive - Paternal Effects - prostate, seminal vesicle, Cowper's gland,
accessory glands
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 30,1079,1985
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 27 gm/kg
- SEX/DURATION :
- female 17-22 day(s) after conception
lactating female 21 day(s) post-birth
- TOXIC EFFECTS :
- Reproductive - Fertility - pre-implantation mortality (e.g. reduction in
number of implants per female; total number of implants per corpora lutea)
Reproductive - Effects on Newborn - viability index (e.g., # alive at day 4
per # born alive)
Reproductive - Effects on Newborn - growth statistics (e.g.%, reduced weight
gain)
- REFERENCE :
- KSRNAM Kiso to Rinsho. Clinical Report. (Yubunsha Co., Ltd., 1-5, Kanda
Suda-Cho, Chiyoda-ku, KS Bldg., Tokyo 101, Japan) V.1- 1960-
Volume(issue)/page/year: 20,6911,1986
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 27 gm/kg
- SEX/DURATION :
- female 17-22 day(s) after conception
lactating female 21 day(s) post-birth
- TOXIC EFFECTS :
- Reproductive - Effects on Newborn - physical
Reproductive - Effects on Newborn - delayed effects
- REFERENCE :
- KSRNAM Kiso to Rinsho. Clinical Report. (Yubunsha Co., Ltd., 1-5, Kanda
Suda-Cho, Chiyoda-ku, KS Bldg., Tokyo 101, Japan) V.1- 1960-
Volume(issue)/page/year: 20,6911,1986
- TYPE OF TEST :
- TCLo - Lowest published toxic concentration
- ROUTE OF EXPOSURE :
- Inhalation
- DOSE :
- 860 ug/m3
- SEX/DURATION :
- female 1-7 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - hepatobiliary system
Reproductive - Effects on Newborn - weaning or lactation index (e.g., #
alive at weaning per # alive at day 4)
Reproductive - Effects on Newborn - growth statistics (e.g.%, reduced weight
gain)
- REFERENCE :
- TOVEFN Toksikologicheskii Vestnik. (18-20 Vadkovskii per. Moscow, 101479,
Russia) History Unknown Volume(issue)/page/year: (5),28,1996
- TYPE OF TEST :
- TCLo - Lowest published toxic concentration
- ROUTE OF EXPOSURE :
- Inhalation
- DOSE :
- 100 ug/m3
- SEX/DURATION :
- female 1-7 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Newborn - delayed effects
- REFERENCE :
- TOVEFN Toksikologicheskii Vestnik. (18-20 Vadkovskii per. Moscow, 101479,
Russia) History Unknown Volume(issue)/page/year: (5),28,1996