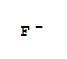Safety
Human poison by ingestion. Human systemic effects by ingestion: convulsions, changes in the respiratory system, liver, and kidneys. Mutation data reported. When heated to decomposition it emits toxic fumes of F−.
Fluoride-containing compounds are so diverse that it is not possible to generalize on their toxicity, which depends on their reactivity and structure, and in the case of salts, their solubility and ability to release fluoride ions.Reaction of the irreversible inhibitor diisopropylfluorophosphate with a serine proteaseSoluble fluoride salts, of which NaF is the most common, are mildly toxic but have resulted in both accidental and suicidal deaths from acute poisoning.While the minimum fatal dose in humans is not known, a case of a fatal poisoning of an adult with 4 grams of NaF is documented.Sodium fluorosilicate For Na2SiF6, the 50% lethal dose (LD50) orally in rats is 0.125 g/kg, corresponding to 12.5 for a 100 kg adult.
. The fatal period ranges from 5 min to 12 hours.The mechanism of toxicity involves the combination of the fluoride anion with the calcium ions in the blood to form insoluble calcium fluoride, resulting in hypocalcemia; calcium is indispensable for the function of the nervous system, and the condition can be fatal. Treatment may involve oral administration of dilute calcium hydroxide or calcium chloride to prevent further absorption, and injection of calcium gluconate to increase the calcium levels in the blood.Hydrogen fluoride is more dangerous than salts such as NaF because it is corrosive and volatile, and can result in fatal exposure through inhalation or upon contact with the skin; calcium gluconate gel is the usual antidote.A few organofluorine compounds are extremely toxic, such as organophosphates like sarin and diisopropylfluorophosphate that react with the cholinesterase enzyme at neuromuscular junctions and thus block the transmission of nerve impulses to the muscles.Here, a reactive fluorine-phosphorus bond in the inhibitor is the site of nucleophilic attack by a serine residue in the enzyme's active site, causing the loss of a F- ion and alkylation and inactivation of the enzyme.While PTFE itself is chemically inert and non-toxic, it begins to deteriorate near or above 500 °F (260 °C), and decompose completely at temperatures above 660 °F (350 °C).These degradation products can be lethal to birds, and can cause flu-like symptoms in humans.In comparison, cooking fats, oils, and butter will begin to scorch and smoke at about 392 °F (200 °C), and meat is usually fried between 400–450 °F (200–230 °C), but empty cookware can exceed this temperature if left unattended on a hot burner.