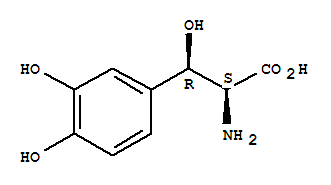
L-Tyrosine, b,3-dihydroxy-, (bR)- cas no:23651-95-8
Synonyms: L-Tyrosine,b,3-dihydroxy-, threo-;Serine,3-(3,4-dihydroxyphenyl)-, L-threo- (8CI);(-)-threo-3,4-Dihydroxyphenylserine;(2S,3R)-3-(3,4-Dihydroxyphenyl)-2-amino-3-hydroxypropanoic acid;DOPS;Droxidopa;L-DOPS;L-Threodops;L-threo-3,4-Dihydroxyphenylserine;L-threo-3-(3,4-Dihydroxyphenyl)serine;L-threo-DOPS;L-threo-b-(3,4-Dihydroxyphenyl)serine;SM5688;threo-Dopaserine;
NameL-Tyrosine, b,3-dihydroxy-, (bR)-
CAS23651-95-8
SynonymsL-Tyrosine,b,3-dihydroxy-, threo-;Serine,3-(3,4-dihydroxyphenyl)-, L-threo- (8CI);(-)-threo-3,4-Dihydroxyphenylserine;(2S,3R)-3-(3,4-Dihydroxyphenyl)-2-amino-3-hydroxypropanoic acid;DOPS;Droxidopa;L-DOPS;L-Threodops;L-threo-3,4-Dihydroxyphenylserine;L-threo-3-(3,4-Dihydroxyphenyl)serine;L-threo-DOPS;L-threo-b-(3,4-Dihydroxyphenyl)serine;SM5688;threo-Dopaserine;
Molecular FormulaC9H11NO5
Molecular Weight213.19
refractive index1.692
CHEMICAL IDENTIFICATION
- RTECS NUMBER :
- VT9626010
- CHEMICAL NAME :
- Serine, 3-(3,4-dihydroxyphenyl)-, L-threo-
- CAS REGISTRY NUMBER :
- 23651-95-8
- LAST UPDATED :
- 199603
- DATA ITEMS CITED :
- 12
- MOLECULAR FORMULA :
- C9-H11-N-O5
- MOLECULAR WEIGHT :
- 213.21
HEALTH HAZARD DATA
ACUTE TOXICITY DATA
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- >10 gm/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- NIIRDN Drugs in Japan (Ethical Drugs). (Yakugyo Jiho Co., Ltd., Tokyo,
Japan) Volume(issue)/page/year: -,758,1990
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 84 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- NIIRDN Drugs in Japan (Ethical Drugs). (Yakugyo Jiho Co., Ltd., Tokyo,
Japan) Volume(issue)/page/year: -,758,1990
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 16 mg/kg
- TOXIC EFFECTS :
- Behavioral - somnolence (general depressed activity)
Behavioral - ataxia
Lungs, Thorax, or Respiration - other changes
- REFERENCE :
- YACHDS Yakuri to Chiryo. Pharmacology and Therapeutics. (Raifu Saiensu
Shuppan K.K., 2-5-13, Yaesu, Chuo-ku, Tokyo 104, Japan) V.1- 1972-
Volume(issue)/page/year: 15(Suppl 2),257,1987
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- >10 gm/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- NIIRDN Drugs in Japan (Ethical Drugs). (Yakugyo Jiho Co., Ltd., Tokyo,
Japan) Volume(issue)/page/year: -,758,1990
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- >10 gm/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- NIIRDN Drugs in Japan (Ethical Drugs). (Yakugyo Jiho Co., Ltd., Tokyo,
Japan) Volume(issue)/page/year: -,758,1990
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- >100 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- NIIRDN Drugs in Japan (Ethical Drugs). (Yakugyo Jiho Co., Ltd., Tokyo,
Japan) Volume(issue)/page/year: -,758,1990
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Mammal - dog
- DOSE/DURATION :
- >5 gm/kg
- TOXIC EFFECTS :
- Cardiac - other changes
Lungs, Thorax, or Respiration - dyspnea
Kidney, Ureter, Bladder - other changes
- REFERENCE :
- YKYUA6 Yakkyoku. Pharmacy. (Nanzando, 4-1-11, Yushima, Bunkyo-ku, Tokyo,
Japan) V.1- 1950- Volume(issue)/page/year: 40,1935,1989
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Primate - monkey
- DOSE/DURATION :
- >5 gm/kg
- TOXIC EFFECTS :
- Cardiac - other changes
Lungs, Thorax, or Respiration - respiratory depression
Kidney, Ureter, Bladder - other changes
- REFERENCE :
- YKYUA6 Yakkyoku. Pharmacy. (Nanzando, 4-1-11, Yushima, Bunkyo-ku, Tokyo,
Japan) V.1- 1950- Volume(issue)/page/year: 40,1935,1989
** REPRODUCTIVE DATA **
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 13200 mg/kg
- SEX/DURATION :
- female 15 day(s) pre-mating
female 1-7 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Maternal Effects - ovaries, fallopian tubes
- REFERENCE :
- YACHDS Yakuri to Chiryo. Pharmacology and Therapeutics. (Raifu Saiensu
Shuppan K.K., 2-5-13, Yaesu, Chuo-ku, Tokyo 104, Japan) V.1- 1972-
Volume(issue)/page/year: 15(Suppl 2),283,1987
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 2200 mg/kg
- SEX/DURATION :
- female 7-17 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Maternal Effects - other effects
Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death,
e.g., stunted fetus)
Reproductive - Specific Developmental Abnormalities - musculoskeletal system
- REFERENCE :
- YACHDS Yakuri to Chiryo. Pharmacology and Therapeutics. (Raifu Saiensu
Shuppan K.K., 2-5-13, Yaesu, Chuo-ku, Tokyo 104, Japan) V.1- 1972-
Volume(issue)/page/year: 15(Suppl 2),283,1987
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 1500 mg/kg
- SEX/DURATION :
- female 17-21 day(s) after conception
lactating female 20 day(s) post-birth
- TOXIC EFFECTS :
- Reproductive - Effects on Newborn - growth statistics (e.g.%, reduced weight
gain)
Reproductive - Effects on Newborn - physical
Reproductive - Effects on Newborn - other postnatal measures or effects
- REFERENCE :
- YACHDS Yakuri to Chiryo. Pharmacology and Therapeutics. (Raifu Saiensu
Shuppan K.K., 2-5-13, Yaesu, Chuo-ku, Tokyo 104, Japan) V.1- 1972-
Volume(issue)/page/year: 15(Suppl 2),283,1987
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 15 gm/kg
- SEX/DURATION :
- female 17-21 day(s) after conception
lactating female 20 day(s) post-birth
- TOXIC EFFECTS :
- Reproductive - Maternal Effects - parturition
- REFERENCE :
- YACHDS Yakuri to Chiryo. Pharmacology and Therapeutics. (Raifu Saiensu
Shuppan K.K., 2-5-13, Yaesu, Chuo-ku, Tokyo 104, Japan) V.1- 1972-
Volume(issue)/page/year: 15(Suppl 2),283,1987