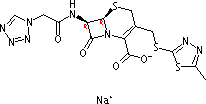
5-Thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylicacid,3-[[(5-methyl-1,3,4-thiadiazol-2-yl)thio]methyl]-8-oxo-7-[[2-(1H-tetrazol-1-yl)acetyl]amino]-,sodium salt (1:1), (6R,7R)- cas no:27164-46-1
Synonyms: 5-Thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylicacid,3-[[(5-methyl-1,3,4-thiadiazol-2-yl)thio]methyl]-8-oxo-7-[(1H-tetrazol-1-ylacetyl)amino]-,monosodium salt, (6R,7R)- (9CI);5-Thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylicacid, 3-[[(5-methyl-1,3,4-thiadiazol-2-yl)thio]methyl]-8-oxo-7-[2-(1H-tetrazol-1-yl)acetamido]-,monosodium salt (8CI);3-[[(5-Methyl-1,3,4-thiadiazol-2-yl)thio]methyl]-8-oxo-7-[2-(1H-tetrazol-1-yl)acetamido]-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylicacid sodium salt;Acef;Ancef;Atirin;Biazolina;Bor-Cefazol;Cefacidal;Cefazil;Cefazoliin;Cefazolin sodium;Cefazoline sodium;Cephazolin sodium;Cephazolin sodiumsalt;Firmacef;Gramaxin;Kefzol;Lampocef;Liviclina;Monosodium cafazolin;Reflin;SKF 41558;Sodium cefazolin;Sodium cephazolin;Totacef;Zolicef;
Name5-Thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylicacid,3-[[(5-methyl-1,3,4-thiadiazol-2-yl)thio]methyl]-8-oxo-7-[[2-(1H-tetrazol-1-yl)acetyl]amino]-,sodium salt (1:1), (6R,7R)-
CAS27164-46-1
Synonyms5-Thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylicacid,3-[[(5-methyl-1,3,4-thiadiazol-2-yl)thio]methyl]-8-oxo-7-[(1H-tetrazol-1-ylacetyl)amino]-,monosodium salt, (6R,7R)- (9CI);5-Thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylicacid, 3-[[(5-methyl-1,3,4-thiadiazol-2-yl)thio]methyl]-8-oxo-7-[2-(1H-tetrazol-1-yl)acetamido]-,monosodium salt (8CI);3-[[(5-Methyl-1,3,4-thiadiazol-2-yl)thio]methyl]-8-oxo-7-[2-(1H-tetrazol-1-yl)acetamido]-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylicacid sodium salt;Acef;Ancef;Atirin;Biazolina;Bor-Cefazol;Cefacidal;Cefazil;Cefazoliin;Cefazolin sodium;Cefazoline sodium;Cephazolin sodium;Cephazolin sodiumsalt;Firmacef;Gramaxin;Kefzol;Lampocef;Liviclina;Monosodium cafazolin;Reflin;SKF 41558;Sodium cefazolin;Sodium cephazolin;Totacef;Zolicef;
EINECS(EC#)248-278-4
Molecular FormulaC14H14N8O4S3. Na
Molecular Weight476.48899
AppearanceYellowish-white odorless, crystalline powder
refractive index20 ° (C=10, H2O)
storage temp2-8°C
Hazard Xn:Harmful
Risk R42/43
CHEMICAL IDENTIFICATION
- RTECS NUMBER :
- XI0390000
- CHEMICAL NAME :
- 5-Thia-1-azabicyclo(4.2.0)oct-2-ene-2-carboxylic
acid, 3-(((5-methyl-1,3,4-thiadiazol-2-yl)
thio)methyl)-8-oxo-7-(((1H-tetrazol-1-yl)acetyl)amino
)-, (6R-trans)-, sodium salt
- CAS REGISTRY NUMBER :
- 27164-46-1
- LAST UPDATED :
- 199706
- DATA ITEMS CITED :
- 28
- MOLECULAR FORMULA :
- C14-H13-N8-O4-S3.Na
- MOLECULAR WEIGHT :
- 476.52
- WISWESSER LINE NOTATION :
- T46 ANV ES GUTJ HVO CMV1- AT5NNNNJ& G1S- CT5NN DSJ E1
&-NA-
HEALTH HAZARD DATA
ACUTE TOXICITY DATA
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Human - woman
- DOSE/DURATION :
- 660 mg/kg/11D-I
- TOXIC EFFECTS :
- Behavioral - convulsions or effect on seizure threshold
- REFERENCE :
- NPRNAY Nephron. (S. Karger Pub., Inc., 79 Fifth Ave., New York, NY 10003)
V.1- 1964- Volume(issue)/page/year: 45,72,1987
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intramuscular
- SPECIES OBSERVED :
- Human
- DOSE/DURATION :
- 14 mg/kg/D
- TOXIC EFFECTS :
- Gastrointestinal - changes in structure or function of salivary glands
Gastrointestinal - nausea or vomiting
Skin and Appendages - dermatitis, allergic (after systemic exposure)
- REFERENCE :
- JMGZAI Japan Medical Gazette. (Tokyo, Japan) V.1-18(?), 1964-81.
Discontinued. Volume(issue)/page/year: 8(8),10,1971
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- >11 gm/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- NKRZAZ Chemotherapy (Tokyo). (Nippon Kagaku Ryoho Gakkai, 2-20-8 Kamiosaki,
Shinagawa-Ku, Tokyo 141, Japan) V.1- 1953- Volume(issue)/page/year:
18,528,1970
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intraperitoneal
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 7400 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- NIIRDN Drugs in Japan (Ethical Drugs). (Yakugyo Jiho Co., Ltd., Tokyo,
Japan) Volume(issue)/page/year: 6,404,1982
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 10 gm/kg
- TOXIC EFFECTS :
- Behavioral - somnolence (general depressed activity)
Behavioral - convulsions or effect on seizure threshold
Gastrointestinal - hypermotility, diarrhea
- REFERENCE :
- JMGZAI Japan Medical Gazette. (Tokyo, Japan) V.1-18(?), 1964-81.
Discontinued. Volume(issue)/page/year: 8(8),10,1971
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 2760 mg/kg
- TOXIC EFFECTS :
- Behavioral - convulsions or effect on seizure threshold
Behavioral - ataxia
Lungs, Thorax, or Respiration - dyspnea
- REFERENCE :
- NKRZAZ Chemotherapy (Tokyo). (Nippon Kagaku Ryoho Gakkai, 2-20-8 Kamiosaki,
Shinagawa-Ku, Tokyo 141, Japan) V.1- 1953- Volume(issue)/page/year:
35(Suppl 1),207,1987
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- >11 gm/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- NKRZAZ Chemotherapy (Tokyo). (Nippon Kagaku Ryoho Gakkai, 2-20-8 Kamiosaki,
Shinagawa-Ku, Tokyo 141, Japan) V.1- 1953- Volume(issue)/page/year:
18,528,1970
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intraperitoneal
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 6200 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- NIIRDN Drugs in Japan (Ethical Drugs). (Yakugyo Jiho Co., Ltd., Tokyo,
Japan) Volume(issue)/page/year: 6,404,1982
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 7600 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- JIDIAQ Journal of Infectious Diseases. (Univ. of Chicago Press, Journals
Div., POB 37005, Chicago, IL 60637) V.1- 1904- Volume(issue)/page/year:
128,S379,1973
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 3900 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- JIDIAQ Journal of Infectious Diseases. (Univ. of Chicago Press, Journals
Div., POB 37005, Chicago, IL 60637) V.1- 1904- Volume(issue)/page/year:
128,S379,1973
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Mammal - dog
- DOSE/DURATION :
- 4 gm/kg
- TOXIC EFFECTS :
- Gastrointestinal - changes in structure or function of salivary glands
Gastrointestinal - hypermotility, diarrhea
Gastrointestinal - nausea or vomiting
- REFERENCE :
- NKRZAZ Chemotherapy (Tokyo). (Nippon Kagaku Ryoho Gakkai, 2-20-8 Kamiosaki,
Shinagawa-Ku, Tokyo 141, Japan) V.1- 1953- Volume(issue)/page/year:
18,528,1970
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Mammal - dog
- DOSE/DURATION :
- 2200 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- ARZNAD Arzneimittel-Forschung. Drug Research. (Editio Cantor Verlag,
Postfach 1255, W-7960 Aulendorf, Fed. Rep. Ger.) V.1- 1951-
Volume(issue)/page/year: 29,424,1979
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - rabbit
- DOSE/DURATION :
- >6 gm/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- NKRZAZ Chemotherapy (Tokyo). (Nippon Kagaku Ryoho Gakkai, 2-20-8 Kamiosaki,
Shinagawa-Ku, Tokyo 141, Japan) V.1- 1953- Volume(issue)/page/year:
18,528,1970
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Rodent - rabbit
- DOSE/DURATION :
- 2500 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- ARZNAD Arzneimittel-Forschung. Drug Research. (Editio Cantor Verlag,
Postfach 1255, W-7960 Aulendorf, Fed. Rep. Ger.) V.1- 1951-
Volume(issue)/page/year: 29,424,1979
** OTHER MULTIPLE DOSE TOXICITY DATA **
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 112 gm/kg/28D-I
- TOXIC EFFECTS :
- Blood - normocytic anemia
Blood - changes in bone marrow (not otherwise specified)
Related to Chronic Data - changes in prostate weight
- REFERENCE :
- JZKEDZ Jitchuken Zenrinsho Kenkyuho. Central Institute for Experimental
Animals, Research Reports. (Jikken Dobutsu Chuo Kenkyusho, 1433 Nogawa,
Takatsu-ku, Kawasaki 211, Japan) V.1- 1975- Volume(issue)/page/year:
2,1,1976
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 91 gm/kg/13W-I
- TOXIC EFFECTS :
- Endocrine - changes in spleen weight
Blood - changes in spleen
Skin and Appendages - dermatitis, other (after systemic exposure)
- REFERENCE :
- NKRZAZ Chemotherapy (Tokyo). (Nippon Kagaku Ryoho Gakkai, 2-20-8 Kamiosaki,
Shinagawa-Ku, Tokyo 141, Japan) V.1- 1953- Volume(issue)/page/year:
18,528,1970
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 21 gm/kg/21D-I
- TOXIC EFFECTS :
- Kidney, Ureter, Bladder - changes in tubules (including acute renal failure,
acute tubular necrosis)
Kidney, Ureter, Bladder - other changes in urine composition
Nutritional and Gross Metabolic - changes in chlorine
- REFERENCE :
- NKRZAZ Chemotherapy (Tokyo). (Nippon Kagaku Ryoho Gakkai, 2-20-8 Kamiosaki,
Shinagawa-Ku, Tokyo 141, Japan) V.1- 1953- Volume(issue)/page/year:
40(Suppl 4),162,1992
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 42875 mg/kg/5W-I
- TOXIC EFFECTS :
- Gastrointestinal - other changes
Nutritional and Gross Metabolic - weight loss or decreased weight gain
- REFERENCE :
- KSRNAM Kiso to Rinsho. Clinical Report. (Yubunsha Co., Ltd., 1-5, Kanda
Suda-Cho, Chiyoda-ku, KS Bldg., Tokyo 101, Japan) V.1- 1960-
Volume(issue)/page/year: 18,1793,1984
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Mammal - dog
- DOSE/DURATION :
- 91 gm/kg/13W-I
- TOXIC EFFECTS :
- Blood - normocytic anemia
Blood - changes in spleen
Skin and Appendages - dermatitis, other (after systemic exposure)
- REFERENCE :
- NKRZAZ Chemotherapy (Tokyo). (Nippon Kagaku Ryoho Gakkai, 2-20-8 Kamiosaki,
Shinagawa-Ku, Tokyo 141, Japan) V.1- 1953- Volume(issue)/page/year:
18,528,1970
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Mammal - dog
- DOSE/DURATION :
- 45500 mg/kg/26W-I
- TOXIC EFFECTS :
- Skin and Appendages - dermatitis, other (after systemic exposure)
- REFERENCE :
- NKRZAZ Chemotherapy (Tokyo). (Nippon Kagaku Ryoho Gakkai, 2-20-8 Kamiosaki,
Shinagawa-Ku, Tokyo 141, Japan) V.1- 1953- Volume(issue)/page/year:
18,528,1970
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Mammal - dog
- DOSE/DURATION :
- 72 gm/kg/13W-I
- TOXIC EFFECTS :
- Liver - changes in liver weight
Blood - normocytic anemia
Biochemical - Enzyme inhibition, induction, or change in blood or tissue
levels - transaminases
- REFERENCE :
- NKRZAZ Chemotherapy (Tokyo). (Nippon Kagaku Ryoho Gakkai, 2-20-8 Kamiosaki,
Shinagawa-Ku, Tokyo 141, Japan) V.1- 1953- Volume(issue)/page/year:
35(Suppl 1),251,1987
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Mammal - dog
- DOSE/DURATION :
- 7500 mg/kg/30D-I
- TOXIC EFFECTS :
- Gastrointestinal - nausea or vomiting
Liver - fatty liver degeneration
Kidney, Ureter, Bladder - other changes
- REFERENCE :
- NKRZAZ Chemotherapy (Tokyo). (Nippon Kagaku Ryoho Gakkai, 2-20-8 Kamiosaki,
Shinagawa-Ku, Tokyo 141, Japan) V.1- 1953- Volume(issue)/page/year:
18,528,1970
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Rodent - rabbit
- DOSE/DURATION :
- 2800 mg/kg/7D-I
- TOXIC EFFECTS :
- Blood - changes in serum composition (e.g. TP, bilirubin, cholesterol)
Biochemical - Enzyme inhibition, induction, or change in blood or tissue
levels - transaminases
Related to Chronic Data - death
- REFERENCE :
- NKRZAZ Chemotherapy (Tokyo). (Nippon Kagaku Ryoho Gakkai, 2-20-8 Kamiosaki,
Shinagawa-Ku, Tokyo 141, Japan) V.1- 1953- Volume(issue)/page/year:
27(Suppl 5),701,1979
** REPRODUCTIVE DATA **
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravenous
- DOSE :
- 5500 mg/kg
- SEX/DURATION :
- female 7-17 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Fertility - post-implantation mortality (e.g. dead and/or
resorbed implants per total number of implants)
Reproductive - Fertility - litter size (e.g. # fetuses per litter; measured
before birth)
Reproductive - Effects on Embryo or Fetus - extra-embryonic structures
(e.g., placenta, umbilical cord)
- REFERENCE :
- NKRZAZ Chemotherapy (Tokyo). (Nippon Kagaku Ryoho Gakkai, 2-20-8 Kamiosaki,
Shinagawa-Ku, Tokyo 141, Japan) V.1- 1953- Volume(issue)/page/year:
28(Suppl 7),1119,1980
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravenous
- DOSE :
- 5500 mg/kg
- SEX/DURATION :
- female 7-17 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death,
e.g., stunted fetus)
Reproductive - Specific Developmental Abnormalities - musculoskeletal system
Reproductive - Effects on Newborn - growth statistics (e.g.%, reduced weight
gain)
- REFERENCE :
- NKRZAZ Chemotherapy (Tokyo). (Nippon Kagaku Ryoho Gakkai, 2-20-8 Kamiosaki,
Shinagawa-Ku, Tokyo 141, Japan) V.1- 1953- Volume(issue)/page/year:
28(Suppl 7),1119,1980
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravenous
- DOSE :
- 21 gm/kg
- SEX/DURATION :
- female 17-22 day(s) after conception
lactating female 20 day(s) post-birth
- TOXIC EFFECTS :
- Reproductive - Maternal Effects - ovaries, fallopian tubes
Reproductive - Maternal Effects - uterus, cervix, vagina
Reproductive - Effects on Newborn - growth statistics (e.g.%, reduced weight
gain)
- REFERENCE :
- NKRZAZ Chemotherapy (Tokyo). (Nippon Kagaku Ryoho Gakkai, 2-20-8 Kamiosaki,
Shinagawa-Ku, Tokyo 141, Japan) V.1- 1953- Volume(issue)/page/year:
35(Suppl 1),404,1987
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravenous
- DOSE :
- 21 gm/kg
- SEX/DURATION :
- female 17-22 day(s) after conception
lactating female 20 day(s) post-birth
- TOXIC EFFECTS :
- Reproductive - Paternal Effects - testes, epididymis, sperm duct
- REFERENCE :
- NKRZAZ Chemotherapy (Tokyo). (Nippon Kagaku Ryoho Gakkai, 2-20-8 Kamiosaki,
Shinagawa-Ku, Tokyo 141, Japan) V.1- 1953- Volume(issue)/page/year:
35(Suppl 1),404,1987
*** NIOSH STANDARDS DEVELOPMENT AND SURVEILLANCE DATA ***
NIOSH OCCUPATIONAL EXPOSURE SURVEY DATA :
NOES - National Occupational Exposure Survey (1983)
NOES Hazard Code - X3195
No. of Facilities: 675 (estimated)
No. of Industries: 1
No. of Occupations: 6
No. of Employees: 27888 (estimated)
No. of Female Employees: 22194 (estimated)