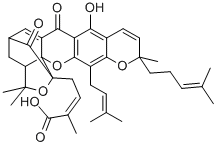
2-Butenoic acid,2-methyl-4-[(1R,3aS,5S,11R,14aS)-3a,4,5,7-tetrahydro-8-hydroxy-3,3,11-trimethyl-13-(3-methyl-2-buten-1-yl)-11-(4-methyl-3-penten-1-yl)-7,15-dioxo-1,5-methano-1H,3H,11H-furo[3,4-g]pyrano[3,2-b]xanthen-1-yl]-,(2Z)- cas no:2752-65-0
Synonyms: 1,5-Methano-1H,3H,11H-furo[3,4-g]pyrano[3,2-b]xanthene-1-crotonicacid, 3a,4,5,7-tetrahydro-8-hydroxy-a,3,3,11-tetramethyl-13-(3-methyl-2-butenyl)-11-(4-methyl-3-pentenyl)-7,15-dioxo-,(Z)- (8CI); 2-Butenoic acid,2-methyl-4-[(1R,3aS,5S,11R,14aS)-3a,4,5,7-tetrahydro-8-hydroxy-3,3,11-trimethyl-13-(3-methyl-2-butenyl)-11-(4-methyl-3-pentenyl)-7,15-dioxo-1,5-methano-1H,3H,11H-furo[3,4-g]pyrano[3,2-b]xanthen-1-yl]-,(2Z)- (9CI); 2-Butenoic acid,2-methyl-4-[3a,4,5,7-tetrahydro-8-hydroxy-3,3,11-trimethyl-13-(3-methyl-2-butenyl)-11-(4-methyl-3-pentenyl)-7,15-dioxo-1,5-methano-1H,3H,11H-furo[3,4-g]pyrano[3,2-b]xanthen-1-yl]-,[1R-[1a,1(Z),3ab,5a,11b,14aS*]]-; Gambogic acid (7CI); R-Gambogic acid; b-Guttiferin
Name2-Butenoic acid,2-methyl-4-[(1R,3aS,5S,11R,14aS)-3a,4,5,7-tetrahydro-8-hydroxy-3,3,11-trimethyl-13-(3-methyl-2-buten-1-yl)-11-(4-methyl-3-penten-1-yl)-7,15-dioxo-1,5-methano-1H,3H,11H-furo[3,4-g]pyrano[3,2-b]xanthen-1-yl]-,(2Z)-
CAS2752-65-0
Synonyms1,5-Methano-1H,3H,11H-furo[3,4-g]pyrano[3,2-b]xanthene-1-crotonicacid, 3a,4,5,7-tetrahydro-8-hydroxy-a,3,3,11-tetramethyl-13-(3-methyl-2-butenyl)-11-(4-methyl-3-pentenyl)-7,15-dioxo-,(Z)- (8CI); 2-Butenoic acid,2-methyl-4-[(1R,3aS,5S,11R,14aS)-3a,4,5,7-tetrahydro-8-hydroxy-3,3,11-trimethyl-13-(3-methyl-2-butenyl)-11-(4-methyl-3-pentenyl)-7,15-dioxo-1,5-methano-1H,3H,11H-furo[3,4-g]pyrano[3,2-b]xanthen-1-yl]-,(2Z)- (9CI); 2-Butenoic acid,2-methyl-4-[3a,4,5,7-tetrahydro-8-hydroxy-3,3,11-trimethyl-13-(3-methyl-2-butenyl)-11-(4-methyl-3-pentenyl)-7,15-dioxo-1,5-methano-1H,3H,11H-furo[3,4-g]pyrano[3,2-b]xanthen-1-yl]-,[1R-[1a,1(Z),3ab,5a,11b,14aS*]]-; Gambogic acid (7CI); R-Gambogic acid; b-Guttiferin
Molecular FormulaC38H44 O8
Molecular Weight628.75
refractive index1.627
storage tempStore at -20°C
Safety
Poison by subcutaneous, intravenous, and intraperitoneal routes. When heated to decomposition it emits acrid smoke and fumes.
| Organism |
Test Type |
Route |
Reported Dose (Normalized Dose) |
Effect |
Source |
| mammal (species unspecified) |
LD50 |
unreported |
55mg/kg (55mg/kg) |
|
"Zhongliu Yanjiu" Cancer Review, Yu, R., et al., eds., Shanghai Science/Technology Publisher,Peop. Rep. China, 1994Vol. -, Pg. 220, 1994. |
| mouse |
LD50 |
subcutaneous |
354mg/kg (354mg/kg) |
|
"CRC Handbook of Antibiotic Compounds," Vols.1- , Berdy, J., Boca Raton, FL, CRC Press, 1980Vol. 8(1), Pg. 331, 1982. |
| rat |
LD50 |
intraperitoneal |
88mg/kg (88mg/kg) |
liver: other changes
|
Indian Journal of Experimental Biology. Vol. 5, Pg. 96, 1967. |
| rat |
LD50 |
intravenous |
107mg/kg (107mg/kg) |
liver: other changes
|
Indian Journal of Experimental Biology. Vol. 5, Pg. 96, 1967. |