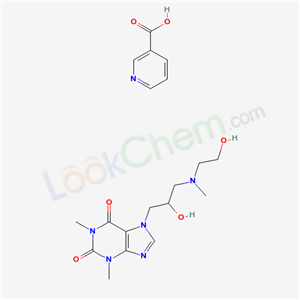
Xanthinol nicotinate cas no:437-74-1
Synonyms: Landrina 300;Complamex;Complamin;7-[2-hydroxy-3-(2-hydroxyethyl-methyl-amino)propyl]-1,3-dimethyl-purine-2,6-dione; pyridine-3-carboxylic acid;3-Pyridinecarboxylic acid, compd. with 3, 7-dihydro-7-[2-hydroxy-3-[ (2-hydroxyethyl)methylamino]propyl]-1, 3-dimethyl-1H-purine-2,6-dione (1:1);Nicotinato de xantinol [INN-Spanish];SK 331A;Complamin retard;Stenalgil;Sadamine;Methoxylin, compd. with nicotinic acid;Vedrin;Xanthinol niacinate [USAN];Sadamin;Xantinoli nicotinas [INN-Latin];
NameXanthinol nicotinate
CAS437-74-1
SynonymsLandrina 300;Complamex;Complamin;7-[2-hydroxy-3-(2-hydroxyethyl-methyl-amino)propyl]-1,3-dimethyl-purine-2,6-dione; pyridine-3-carboxylic acid;3-Pyridinecarboxylic acid, compd. with 3, 7-dihydro-7-[2-hydroxy-3-[ (2-hydroxyethyl)methylamino]propyl]-1, 3-dimethyl-1H-purine-2,6-dione (1:1);Nicotinato de xantinol [INN-Spanish];SK 331A;Complamin retard;Stenalgil;Sadamine;Methoxylin, compd. with nicotinic acid;Vedrin;Xanthinol niacinate [USAN];Sadamin;Xantinoli nicotinas [INN-Latin];
EINECS(EC#)207-115-7
Molecular FormulaC13H21N5O4•C6H5NO2
Molecular Weight434.44634
AppearanceWhite crystalline powder
Globally Harmonized System of Classification adn Labelling of Chemicals(GHS)
HazardclassIRRITANT
Safety Moderately toxic by intraperitoneal and intravenous routes. Mildly toxic by ingestion. An experimental teratogen. Experimental reproductive effects. When heated to decomposition it emits toxic fumes of NOx. Used as a peripheral vasodilator. See also NICOTINIC ACID, BUTYL ESTER; NICOTINIC ACID, 3-(2,6-DIMETHYLPIPERID-INO)PROPYL ESTER, HYDROCHLORIDE; NICOTINIC ACID, HYDRAZIDE; NICOTINIC ACID, METHYL ESTER; NICOTINIC ACID, 3-(2-METHYLPIPERIDINO)- PROPYL ESTER, HYDROCHLORIDE; NICOTINIC ACID, SODIUM SALT.
CHEMICAL IDENTIFICATION
- RTECS NUMBER :
- QT1500000
- CHEMICAL NAME :
- Nicotinic acid, compd. with
3,7-dihydro-7-(2-hydroxy-3-((2-hydroxyethyl)
methylamino)propyl)-1,3-dimethyl-1H-purine-2,6-dione
- CAS REGISTRY NUMBER :
- 437-74-1
- LAST UPDATED :
- 199706
- DATA ITEMS CITED :
- 18
- MOLECULAR FORMULA :
- C13-H21-N5-O4.C6-H5-N-O2
- MOLECULAR WEIGHT :
- 434.51
- WISWESSER LINE NOTATION :
- T56 BN DN FNVNVJ B1YQ1N1&2Q F1 H1 &T6NJ CVQ
HEALTH HAZARD DATA
ACUTE TOXICITY DATA
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 14130 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 9,601,1975
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intraperitoneal
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 3010 mg/kg
- TOXIC EFFECTS :
- Behavioral - altered sleep time (including change in righting reflex)
Lungs, Thorax, or Respiration - cyanosis
Nutritional and Gross Metabolic - body temperature decrease
- REFERENCE :
- TOIZAG Toho Igakkai Zasshi. Journal of Medical Society of Toho University.
(Toho Daigaku Igakkai, 21-16, Omori-nishi, 5-chome, Ota-ku, Tokyo 143,
Japan) V.1- 1954- Volume(issue)/page/year: 15,424,1968
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 4061 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- TOIZAG Toho Igakkai Zasshi. Journal of Medical Society of Toho University.
(Toho Daigaku Igakkai, 21-16, Omori-nishi, 5-chome, Ota-ku, Tokyo 143,
Japan) V.1- 1954- Volume(issue)/page/year: 15,424,1968
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 690 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 9,601,1975
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 17350 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 9,601,1975
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 4260 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 9,601,1975
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 673 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- YHTPAD Yaoxue Tongbao. Bulletin of Pharmacology. (China International Book
Trading Corp., POB 2820, Beijing, Peop. Rep. China) V.13-23, 1978-88. For
publisher information, see ZYZAEU. Volume(issue)/page/year: 21,291,1986
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Rodent - rabbit
- DOSE/DURATION :
- 500 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 9,601,1975
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Mammal - species unspecified
- DOSE/DURATION :
- 900 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- PCJOAU Pharmaceutical Chemistry Journal (English Translation). Translation
of KHFZAN. (Plenum Pub. Corp., 233 Spring St., New York, NY 10013) No.1-
1967- Volume(issue)/page/year: 20,286,1986
** OTHER MULTIPLE DOSE TOXICITY DATA **
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 749 gm/kg/26W-I
- TOXIC EFFECTS :
- Liver - other changes
Related to Chronic Data - death
- REFERENCE :
- TOIZAG Toho Igakkai Zasshi. Journal of Medical Society of Toho University.
(Toho Daigaku Igakkai, 21-16, Omori-nishi, 5-chome, Ota-ku, Tokyo 143,
Japan) V.1- 1954- Volume(issue)/page/year: 18,317,1971
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intraperitoneal
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 9880 mg/kg/30D-I
- TOXIC EFFECTS :
- Related to Chronic Data - death
- REFERENCE :
- TOIZAG Toho Igakkai Zasshi. Journal of Medical Society of Toho University.
(Toho Daigaku Igakkai, 21-16, Omori-nishi, 5-chome, Ota-ku, Tokyo 143,
Japan) V.1- 1954- Volume(issue)/page/year: 18,317,1971
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intraperitoneal
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 117 gm/kg/26W-I
- TOXIC EFFECTS :
- Liver - other changes
Kidney, Ureter, Bladder - other changes
Related to Chronic Data - death
- REFERENCE :
- TOIZAG Toho Igakkai Zasshi. Journal of Medical Society of Toho University.
(Toho Daigaku Igakkai, 21-16, Omori-nishi, 5-chome, Ota-ku, Tokyo 143,
Japan) V.1- 1954- Volume(issue)/page/year: 18,317,1971
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 13182 mg/kg/30D-I
- TOXIC EFFECTS :
- Related to Chronic Data - death
- REFERENCE :
- TOIZAG Toho Igakkai Zasshi. Journal of Medical Society of Toho University.
(Toho Daigaku Igakkai, 21-16, Omori-nishi, 5-chome, Ota-ku, Tokyo 143,
Japan) V.1- 1954- Volume(issue)/page/year: 18,317,1971
** REPRODUCTIVE DATA **
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 1920 mg/kg
- SEX/DURATION :
- female 7-14 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death,
e.g., stunted fetus)
Reproductive - Effects on Newborn - growth statistics (e.g.%, reduced weight
gain)
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 8,1145,1974
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- DOSE :
- 1920 mg/kg
- SEX/DURATION :
- female 7-14 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death,
e.g., stunted fetus)
Reproductive - Effects on Newborn - growth statistics (e.g.%, reduced weight
gain)
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 8,1145,1974
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- DOSE :
- 10 gm/kg
- SEX/DURATION :
- female 7-14 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Newborn - weaning or lactation index (e.g., #
alive at weaning per # alive at day 4)
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 8,1145,1974
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 1920 mg/kg
- SEX/DURATION :
- female 7-14 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death,
e.g., stunted fetus)
Reproductive - Specific Developmental Abnormalities - musculoskeletal system
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 8,1145,1974
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 40 gm/kg
- SEX/DURATION :
- female 7-14 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Fertility - litter size (e.g. # fetuses per litter; measured
before birth)
Reproductive - Effects on Newborn - growth statistics (e.g.%, reduced weight
gain)
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 8,1145,1974