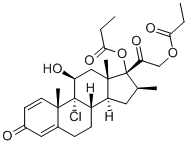
Pregna-1,4-diene-3,20-dione,9-chloro-11-hydroxy-16-methyl-17,21-bis(1-oxopropoxy)-, (11b,16b)- cas no:5534-09-8
Synonyms: 9-Chloro-11b,17,21-trihydroxy-16b-methylpregna-1,4-diene-3,20-dione 17,21-dipropionate;Aldecin;
NamePregna-1,4-diene-3,20-dione,9-chloro-11-hydroxy-16-methyl-17,21-bis(1-oxopropoxy)-, (11b,16b)-
CAS5534-09-8
Synonyms9-Chloro-11b,17,21-trihydroxy-16b-methylpregna-1,4-diene-3,20-dione 17,21-dipropionate;Aldecin;
EINECS(EC#)226-886-0
Molecular FormulaC28H37ClO7
Molecular Weight521.04218
AppearanceWhite crystalline powder
refractive index1.564
storage temp2-8°C
Hazard T:Toxic
Risk R60;R61
Safety Hazard Codes:T,Xn
Risk Statements:60-61-36/37/38-20/21/22
60:May impair fertility
61:May cause harm to the unborn child
36/37/38:Irritating to eyes, respiratory system and skin
20/21/22:Harmful by inhalation, in contact with skin and if swallowed
Safety Statements:53-36/37/39-45-36-26
53:Avoid exposure - obtain special instruction before use
36/37/39:Wear suitable protective clothing, gloves and eye/face
protection
45:In case of accident or if you feel unwell, seek medical
advice immediately (show label where possible)
36:Wear suitable protective clothing
26:In case of contact with eyes, rinse immediately with plenty
of water and seek medical advice
WGK Germany:3
HS Code:29372100
HS Code 29372100
CHEMICAL IDENTIFICATION
- RTECS NUMBER :
- TU3805000
- CHEMICAL NAME :
- Pregna-1-4-diene-3,20-dione,
9-chloro-16-beta-methyl-11-beta,17,21-trihydroxy-,
17,21-dipropionate
- CAS REGISTRY NUMBER :
- 5534-09-8
- LAST UPDATED :
- 199709
- DATA ITEMS CITED :
- 50
- MOLECULAR FORMULA :
- C28-H37-Cl-O7
- MOLECULAR WEIGHT :
- 521.10
- WISWESSER LINE NOTATION :
- L E5 B666 OV AHTTT&J A1 BG CQ E1 FV1Q GQ
HEALTH HAZARD DATA
ACUTE TOXICITY DATA
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- >3750 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 12,863,1976
- TYPE OF TEST :
- LC50 - Lethal concentration, 50 percent kill
- ROUTE OF EXPOSURE :
- Inhalation
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- >51600 ug/m3/2H
- TOXIC EFFECTS :
- Behavioral - convulsions or effect on seizure threshold
Behavioral - muscle contraction or spasticity
- REFERENCE :
- JZKEDZ Jitchuken Zenrinsho Kenkyuho. Central Institute for Experimental
Animals, Research Reports. (Jikken Dobutsu Chuo Kenkyusho, 1433 Nogawa,
Takatsu-ku, Kawasaki 211, Japan) V.1- 1975- Volume(issue)/page/year:
2,97,1976
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intraperitoneal
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- >3 gm/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 12,863,1976
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- >1500 mg/kg
- TOXIC EFFECTS :
- Skin and Appendages - dermatitis, other (after systemic exposure)
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 12,863,1976
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- >5 gm/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- NIIRDN Drugs in Japan (Ethical Drugs). (Yakugyo Jiho Co., Ltd., Tokyo,
Japan) Volume(issue)/page/year: 6,746,1982
- TYPE OF TEST :
- LC50 - Lethal concentration, 50 percent kill
- ROUTE OF EXPOSURE :
- Inhalation
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- >2880 ug/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- NIIRDN Drugs in Japan (Ethical Drugs). (Yakugyo Jiho Co., Ltd., Tokyo,
Japan) Volume(issue)/page/year: 6,746,1982
- TYPE OF TEST :
- LDLo - Lowest published lethal dose
- ROUTE OF EXPOSURE :
- Intraperitoneal
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 2 gm/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- KSRNAM Kiso to Rinsho. Clinical Report. (Yubunsha Co., Ltd., 1-5, Kanda
Suda-Cho, Chiyoda-ku, KS Bldg., Tokyo 101, Japan) V.1- 1960-
Volume(issue)/page/year: 5,1037,1971
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- >5 gm/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- NIIRDN Drugs in Japan (Ethical Drugs). (Yakugyo Jiho Co., Ltd., Tokyo,
Japan) Volume(issue)/page/year: 6,746,1982
- TYPE OF TEST :
- LD - Lethal dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - rabbit
- DOSE/DURATION :
- >750 mg/kg
- TOXIC EFFECTS :
- Liver - other changes
Kidney, Ureter, Bladder - other changes
- REFERENCE :
- YIKUAO Yamaguchi Igaku. Yamaguchi Medicine. (Yamaguchi Daigaku Igakkai,
Kogushi Ube, Yamaguchi- Ken, Japan) V.1- 1953- Volume(issue)/page/year:
19,65,1970
** OTHER MULTIPLE DOSE TOXICITY DATA **
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 960 mg/kg/30D-C
- TOXIC EFFECTS :
- Endocrine - changes in adrenal weight
Endocrine - changes in thymus weight
Nutritional and Gross Metabolic - weight loss or decreased weight gain
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 12,863,1976
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 29120 ug/kg/26W-C
- TOXIC EFFECTS :
- Endocrine - changes in spleen weight
Endocrine - changes in thymus weight
Related to Chronic Data - changes in uterine weight
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 12,877,1976
- TYPE OF TEST :
- TCLo - Lowest published toxic concentration
- ROUTE OF EXPOSURE :
- Inhalation
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 590 mg/m3/26W-C
- TOXIC EFFECTS :
- Endocrine - changes in spleen weight
Endocrine - changes in thymus weight
Blood - changes in leukocyte (WBC) count
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 13,17,1977
- TYPE OF TEST :
- TCLo - Lowest published toxic concentration
- ROUTE OF EXPOSURE :
- Inhalation
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 145 mg/kg/13W-C
- TOXIC EFFECTS :
- Endocrine - changes in spleen weight
Endocrine - changes in thymus weight
Blood - changes in leukocyte (WBC) count
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 13,33,1977
- TYPE OF TEST :
- TCLo - Lowest published toxic concentration
- ROUTE OF EXPOSURE :
- Inhalation
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 80 gm/m3/3M/30D-I
- TOXIC EFFECTS :
- Endocrine - changes in thymus weight
Blood - changes in serum composition (e.g. TP, bilirubin, cholesterol)
Blood - changes in erythrocyte (RBC) count
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 13,1,1977
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Administration onto the skin
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 1875 ug/kg/30D-I
- TOXIC EFFECTS :
- Blood - changes in bone marrow (not otherwise specified)
- REFERENCE :
- JTSCDR Journal of Toxicological Sciences. (Japanese Soc. of Toxicological
Sciences, 4th Floor, Gakkai Center Bldg., 4-16, Yayoi 2-chome, Bunkyo-ku,
Tokyo 113, Japan) V.1- 1976- Volume(issue)/page/year: 7(Suppl
1),15,1982
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 27 gm/kg/13W-I
- TOXIC EFFECTS :
- Blood - changes in erythrocyte (RBC) count
Nutritional and Gross Metabolic - weight loss or decreased weight gain
Biochemical - Enzyme inhibition, induction, or change in blood or tissue
levels - transaminases
- REFERENCE :
- YIKUAO Yamaguchi Igaku. Yamaguchi Medicine. (Yamaguchi Daigaku Igakkai,
Kogushi Ube, Yamaguchi- Ken, Japan) V.1- 1953- Volume(issue)/page/year:
19,65,1970
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 1800 mg/kg/26W-I
- TOXIC EFFECTS :
- Nutritional and Gross Metabolic - weight loss or decreased weight gain
- REFERENCE :
- YIKUAO Yamaguchi Igaku. Yamaguchi Medicine. (Yamaguchi Daigaku Igakkai,
Kogushi Ube, Yamaguchi- Ken, Japan) V.1- 1953- Volume(issue)/page/year:
19,65,1970
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Administration onto the skin
- SPECIES OBSERVED :
- Rodent - rabbit
- DOSE/DURATION :
- 10500 mg/kg/42D-I
- TOXIC EFFECTS :
- Gastrointestinal - ulceration or bleeding from stomach
Gastrointestinal - hypermotility, diarrhea
Related to Chronic Data - death
- REFERENCE :
- YIKUAO Yamaguchi Igaku. Yamaguchi Medicine. (Yamaguchi Daigaku Igakkai,
Kogushi Ube, Yamaguchi- Ken, Japan) V.1- 1953- Volume(issue)/page/year:
19,65,1970
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Administration onto the skin
- SPECIES OBSERVED :
- Rodent - rabbit
- DOSE/DURATION :
- 9 gm/kg/26W-I
- TOXIC EFFECTS :
- Blood - changes in serum composition (e.g. TP, bilirubin, cholesterol)
Blood - changes in other cell count (unspecified)
Biochemical - Enzyme inhibition, induction, or change in blood or tissue
levels - transaminases
- REFERENCE :
- YIKUAO Yamaguchi Igaku. Yamaguchi Medicine. (Yamaguchi Daigaku Igakkai,
Kogushi Ube, Yamaguchi- Ken, Japan) V.1- 1953- Volume(issue)/page/year:
19,65,1970
** REPRODUCTIVE DATA **
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 16 mg/kg
- SEX/DURATION :
- male 9 week(s) pre-mating
female 2 week(s) pre-mating - 7 day(s) after
conception
- TOXIC EFFECTS :
- Reproductive - Fertility - female fertility index (e.g. # females pregnant
per # sperm positive females; # females pregnant per # females mated)
Reproductive - Effects on Embryo or Fetus - extra-embryonic structures
(e.g., placenta, umbilical cord)
Reproductive - Specific Developmental Abnormalities - musculoskeletal system
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 18,1003,1979
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 176 mg/kg
- SEX/DURATION :
- female 7-17 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - endocrine system
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 18,1021,1979
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 1600 ug/kg
- SEX/DURATION :
- male 9 week(s) pre-mating
female 2 week(s) pre-mating - 7 day(s) after
conception
- TOXIC EFFECTS :
- Reproductive - Fertility - pre-implantation mortality (e.g. reduction in
number of implants per female; total number of implants per corpora lutea)
Reproductive - Fertility - litter size (e.g. # fetuses per litter; measured
before birth)
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 18,1003,1979
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 160 ug/kg
- SEX/DURATION :
- male 9 week(s) pre-mating
female 2 week(s) pre-mating - 7 day(s) after
conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death,
e.g., stunted fetus)
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 18,1003,1979
- TYPE OF TEST :
- TCLo - Lowest published toxic concentration
- ROUTE OF EXPOSURE :
- Inhalation
- DOSE :
- 24 ug/kg/3M
- SEX/DURATION :
- female 8-17 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death,
e.g., stunted fetus)
Reproductive - Specific Developmental Abnormalities - musculoskeletal system
Reproductive - Effects on Newborn - weaning or lactation index (e.g., #
alive at weaning per # alive at day 4)
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 13,185,1977
- TYPE OF TEST :
- TCLo - Lowest published toxic concentration
- ROUTE OF EXPOSURE :
- Inhalation
- DOSE :
- 24 ug/kg/3M
- SEX/DURATION :
- female 8-17 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Newborn - physical
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 13,185,1977
- TYPE OF TEST :
- TCLo - Lowest published toxic concentration
- ROUTE OF EXPOSURE :
- Inhalation
- DOSE :
- 168 ug/kg/3M
- SEX/DURATION :
- female 8-17 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Newborn - growth statistics (e.g.%, reduced weight
gain)
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 13,185,1977
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- DOSE :
- 84 gm/kg
- SEX/DURATION :
- female 4 week(s) pre-mating
- TOXIC EFFECTS :
- Reproductive - Maternal Effects - uterus, cervix, vagina
Reproductive - Maternal Effects - menstrual cycle changes or disorders
- REFERENCE :
- JZKEDZ Jitchuken Zenrinsho Kenkyuho. Central Institute for Experimental
Animals, Research Reports. (Jikken Dobutsu Chuo Kenkyusho, 1433 Nogawa,
Takatsu-ku, Kawasaki 211, Japan) V.1- 1975- Volume(issue)/page/year:
2,237,1976
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 720 ug/kg
- SEX/DURATION :
- female 7-15 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - musculoskeletal system
Reproductive - Effects on Newborn - growth statistics (e.g.%, reduced weight
gain)
Reproductive - Effects on Newborn - physical
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 13,175,1977
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 7200 ug/kg
- SEX/DURATION :
- female 7-15 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Maternal Effects - parturition
Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death,
e.g., stunted fetus)
Reproductive - Specific Developmental Abnormalities - craniofacial
(including nose and tongue)
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 13,175,1977
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 72 mg/kg
- SEX/DURATION :
- female 7-15 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetal death
Reproductive - Effects on Newborn - live birth index (measured after birth)
Reproductive - Effects on Newborn - sex ratio
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 13,175,1977
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 576 mg/kg
- SEX/DURATION :
- female 7-15 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Fertility - post-implantation mortality (e.g. dead and/or
resorbed implants per total number of implants)
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 13,175,1977
- TYPE OF TEST :
- TCLo - Lowest published toxic concentration
- ROUTE OF EXPOSURE :
- Inhalation
- DOSE :
- 11 ug/kg/3M
- SEX/DURATION :
- female 7-15 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death,
e.g., stunted fetus)
Reproductive - Specific Developmental Abnormalities - musculoskeletal system
Reproductive - Specific Developmental Abnormalities - other developmental
abnormalities
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 13,195,1977
- TYPE OF TEST :
- TCLo - Lowest published toxic concentration
- ROUTE OF EXPOSURE :
- Inhalation
- DOSE :
- 11 ug/kg/3M
- SEX/DURATION :
- female 7-15 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Newborn - growth statistics (e.g.%, reduced weight
gain)
Reproductive - Effects on Newborn - physical
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 13,195,1977
- TYPE OF TEST :
- TCLo - Lowest published toxic concentration
- ROUTE OF EXPOSURE :
- Inhalation
- DOSE :
- 17 mg/m3/5M
- SEX/DURATION :
- male 9 week(s) pre-mating
female 2 week(s) pre-mating - 6 day(s) after
conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death,
e.g., stunted fetus)
Reproductive - Effects on Embryo or Fetus - fetal death
Reproductive - Specific Developmental Abnormalities - musculoskeletal system
- REFERENCE :
- JZKEDZ Jitchuken Zenrinsho Kenkyuho. Central Institute for Experimental
Animals, Research Reports. (Jikken Dobutsu Chuo Kenkyusho, 1433 Nogawa,
Takatsu-ku, Kawasaki 211, Japan) V.1- 1975- Volume(issue)/page/year:
2,201,1976
- TYPE OF TEST :
- TCLo - Lowest published toxic concentration
- ROUTE OF EXPOSURE :
- Inhalation
- DOSE :
- 58 ug/kg/3M
- SEX/DURATION :
- female 7-15 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Newborn - weaning or lactation index (e.g., #
alive at weaning per # alive at day 4)
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 13,195,1977
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- DOSE :
- 700 ug/kg
- SEX/DURATION :
- female 7-13 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death,
e.g., stunted fetus)
Reproductive - Specific Developmental Abnormalities - craniofacial
(including nose and tongue)
Reproductive - Specific Developmental Abnormalities - musculoskeletal system
- REFERENCE :
- YIKUAO Yamaguchi Igaku. Yamaguchi Medicine. (Yamaguchi Daigaku Igakkai,
Kogushi Ube, Yamaguchi- Ken, Japan) V.1- 1953- Volume(issue)/page/year:
19,65,1970
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- DOSE :
- 2100 ug/kg
- SEX/DURATION :
- female 7-13 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetal death
Reproductive - Effects on Newborn - weaning or lactation index (e.g., #
alive at weaning per # alive at day 4)
- REFERENCE :
- YIKUAO Yamaguchi Igaku. Yamaguchi Medicine. (Yamaguchi Daigaku Igakkai,
Kogushi Ube, Yamaguchi- Ken, Japan) V.1- 1953- Volume(issue)/page/year:
19,65,1970
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- DOSE :
- 10 mg/kg
- SEX/DURATION :
- female 6-15 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - extra-embryonic structures
(e.g., placenta, umbilical cord)
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 24,741,1982
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- DOSE :
- 700 ug/kg
- SEX/DURATION :
- female 7-13 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death,
e.g., stunted fetus)
Reproductive - Specific Developmental Abnormalities - musculoskeletal system
- REFERENCE :
- YIKUAO Yamaguchi Igaku. Yamaguchi Medicine. (Yamaguchi Daigaku Igakkai,
Kogushi Ube, Yamaguchi- Ken, Japan) V.1- 1953- Volume(issue)/page/year:
19,65,1970
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- DOSE :
- 2100 ug/kg
- SEX/DURATION :
- female 7-13 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetal death
Reproductive - Specific Developmental Abnormalities - musculoskeletal system
- REFERENCE :
- YIKUAO Yamaguchi Igaku. Yamaguchi Medicine. (Yamaguchi Daigaku Igakkai,
Kogushi Ube, Yamaguchi- Ken, Japan) V.1- 1953- Volume(issue)/page/year:
19,65,1970
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 1920 ug/kg
- SEX/DURATION :
- female 7-18 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Fertility - post-implantation mortality (e.g. dead and/or
resorbed implants per total number of implants)
Reproductive - Effects on Embryo or Fetus - fetal death
Reproductive - Specific Developmental Abnormalities - musculoskeletal system
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 13,71,1977
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 480 ug/kg
- SEX/DURATION :
- female 7-18 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death,
e.g., stunted fetus)
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 13,71,1977
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 960 ug/kg
- SEX/DURATION :
- female 7-18 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - craniofacial
(including nose and tongue)
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 13,71,1977
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 4800 ug/kg
- SEX/DURATION :
- female 7-18 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - skin and skin
appendages
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 13,71,1977
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- DOSE :
- 250 ug/kg
- SEX/DURATION :
- female 7-16 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetal death
Reproductive - Specific Developmental Abnormalities - Central Nervous System
Reproductive - Specific Developmental Abnormalities - craniofacial
(including nose and tongue)
- REFERENCE :
- YIKUAO Yamaguchi Igaku. Yamaguchi Medicine. (Yamaguchi Daigaku Igakkai,
Kogushi Ube, Yamaguchi- Ken, Japan) V.1- 1953- Volume(issue)/page/year:
19,65,1970
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- DOSE :
- 1 mg/kg
- SEX/DURATION :
- female 7-16 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - body wall
- REFERENCE :
- YIKUAO Yamaguchi Igaku. Yamaguchi Medicine. (Yamaguchi Daigaku Igakkai,
Kogushi Ube, Yamaguchi- Ken, Japan) V.1- 1953- Volume(issue)/page/year:
19,65,1970
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- DOSE :
- 250 ug/kg
- SEX/DURATION :
- female 7-16 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetal death
Reproductive - Specific Developmental Abnormalities - Central Nervous System
Reproductive - Specific Developmental Abnormalities - musculoskeletal system
- REFERENCE :
- YIKUAO Yamaguchi Igaku. Yamaguchi Medicine. (Yamaguchi Daigaku Igakkai,
Kogushi Ube, Yamaguchi- Ken, Japan) V.1- 1953- Volume(issue)/page/year:
19,65,1970
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- DOSE :
- 1 mg/kg
- SEX/DURATION :
- female 7-16 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - other developmental
abnormalities
- REFERENCE :
- YIKUAO Yamaguchi Igaku. Yamaguchi Medicine. (Yamaguchi Daigaku Igakkai,
Kogushi Ube, Yamaguchi- Ken, Japan) V.1- 1953- Volume(issue)/page/year:
19,65,1970
*** REVIEWS ***
TOXICOLOGY REVIEW
ARTODN Archives of Toxicology. (Springer-Verlag, Heidelberger Pl. 3, D-1000
Berlin 33, Fed. Rep. Ger.) V.32- 1974- Volume(issue)/page/year:
69,509,1995
*** NIOSH STANDARDS DEVELOPMENT AND SURVEILLANCE DATA ***
NIOSH OCCUPATIONAL EXPOSURE SURVEY DATA :
NOES - National Occupational Exposure Survey (1983)
NOES Hazard Code - X5586
No. of Facilities: 19 (estimated)
No. of Industries: 1
No. of Occupations: 1
No. of Employees: 506 (estimated)
No. of Female Employees: 285 (estimated)