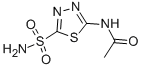
acetazolamide cas no:59-66-5
Synonyms: 2-acetamido-5-sulfamoyl-1,3,4-thiadiazole; 2-acetylamino-1,3,4-thiadiazole-5-sulfonamide; 5-acetamido-1,3,4-thiadiazole-2-sulfonamide
Nameacetazolamide
CAS59-66-5
Synonyms2-acetamido-5-sulfamoyl-1,3,4-thiadiazole; 2-acetylamino-1,3,4-thiadiazole-5-sulfonamide; 5-acetamido-1,3,4-thiadiazole-2-sulfonamide
EINECS(EC#)200-440-5
Molecular FormulaC4H6N4O3S2
Molecular Weight222.24
Appearancewhite to off-white crystalline powder
storage tempStore between 15 and 30C (59 and 86F), in a well-closed container, unless otherwise specified by manufacturer.
Globally Harmonized System of Classification adn Labelling of Chemicals(GHS)
Hazardclass6.1
Hazard Xi:Irritant
Risk R36/38
Safety Hazard Codes:Xi
Risk Statements:36/38-36/37/38
36/38:Irritating to eyes and skin
36/37/38:Irritating to eyes, respiratory system and skin
Safety Statements:26-36
26:In case of contact with eyes, rinse immediately with plenty
of water and seek medical advice
36:Wear suitable protective clothing
RIDADR:2811
HazardClass:6.1
PackingGroup:III
Hazardous Substances Data:59-66-5(Hazardous Substances Data)
CHEMICAL IDENTIFICATION
- RTECS NUMBER :
- AC8225000
- CHEMICAL NAME :
- Acetamide, N-(5-sulfamoyl-1,3,4-thiadiazol-2-yl)-
- CAS REGISTRY NUMBER :
- 59-66-5
- LAST UPDATED :
- 199701
- DATA ITEMS CITED :
- 39
- MOLECULAR FORMULA :
- C4-H6-N4-O3-S2
- MOLECULAR WEIGHT :
- 222.26
- WISWESSER LINE NOTATION :
- T5NN DSJ CSZW EMV1
HEALTH HAZARD DATA
ACUTE TOXICITY DATA
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Human - man
- DOSE/DURATION :
- 54 mg/kg/5D-I
- TOXIC EFFECTS :
- Lungs, Thorax, or Respiration - dyspnea
- REFERENCE :
- AIMDAP Archives of Internal Medicine. (AMA, 535 N. Dearborn St., Chicago,
IL 60610) V.1- 1908- Volume(issue)/page/year: 143,1278,1983
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intraperitoneal
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 2750 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- NYKZAU Nippon Yakurigaku Zasshi. Japanese Journal of Pharmacology. (Nippon
Yakuri Gakkai, c/o Kyoto Daigaku Igakubu Yakurigaku Kyoshitsu, Konoe-cho,
Yoshida, Sakyo-ku, Kyoto 606, Japan) V.40- 1944- Volume(issue)/page/year:
56(4),134S,1960
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 4300 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- ABMGAJ Acta Biologica et Medica Germanica. (Berlin, Ger. Dem. Rep.) V.1-41,
1958-82. For publisher information, see BBIADT. Volume(issue)/page/year:
21,193,1968
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intraperitoneal
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 1175 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- RPTOAN Russian Pharmacology and Toxicology (English Translation).
Translation of FATOAO. (Euromed Pub., 33, Woodlands Rd., Surbiton, Surrey,
UK) V.30- 1967- Volume(issue)/page/year: 39,255,1976
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- >3 gm/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- NIIRDN Drugs in Japan (Ethical Drugs). (Yakugyo Jiho Co., Ltd., Tokyo,
Japan) Volume(issue)/page/year: 6,15,1982
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- >3 gm/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- NIIRDN Drugs in Japan (Ethical Drugs). (Yakugyo Jiho Co., Ltd., Tokyo,
Japan) Volume(issue)/page/year: 6,15,1982
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Mammal - dog
- DOSE/DURATION :
- >2 gm/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- NIIRDN Drugs in Japan (Ethical Drugs). (Yakugyo Jiho Co., Ltd., Tokyo,
Japan) Volume(issue)/page/year: 6,15,1982
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - guinea pig
- DOSE/DURATION :
- >1500 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- NIIRDN Drugs in Japan (Ethical Drugs). (Yakugyo Jiho Co., Ltd., Tokyo,
Japan) Volume(issue)/page/year: 6,15,1982
** REPRODUCTIVE DATA **
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 6600 mg/kg
- SEX/DURATION :
- female 1-22 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - musculoskeletal system
- REFERENCE :
- JHMJAX Johns Hopkins Medical Journal. (Baltimore, MD) V.120-151, 1967-82.
Discontinued. Volume(issue)/page/year: 130,105,1972
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 240 mg/kg
- SEX/DURATION :
- female 9-10 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - musculoskeletal system
- REFERENCE :
- BJEPA5 British Journal of Experimental Pathology. (Blackwell Scientific
Pub. Ltd., POB 88, Oxford, UK) V.1- 1920- Volume(issue)/page/year:
53,5,1972
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 3300 mg/kg
- SEX/DURATION :
- female 1-22 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Fertility - post-implantation mortality (e.g. dead and/or
resorbed implants per total number of implants)
Reproductive - Effects on Embryo or Fetus - fetal death
- REFERENCE :
- TJADAB Teratology, The International Journal of Abnormal Development. (Alan
R. Liss, Inc., 41 E. 11th St., New York, NY 10003) V.1- 1968-
Volume(issue)/page/year: 1,51,1968
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 1500 mg/kg
- SEX/DURATION :
- female 5-7 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetal death
Reproductive - Effects on Newborn - sex ratio
- REFERENCE :
- TJADAB Teratology, The International Journal of Abnormal Development. (Alan
R. Liss, Inc., 41 E. 11th St., New York, NY 10003) V.1- 1968-
Volume(issue)/page/year: 34,452,1986
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 1 gm/kg
- SEX/DURATION :
- female 10 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - extra-embryonic structures
(e.g., placenta, umbilical cord)
- REFERENCE :
- TJADAB Teratology, The International Journal of Abnormal Development. (Alan
R. Liss, Inc., 41 E. 11th St., New York, NY 10003) V.1- 1968-
Volume(issue)/page/year: 34,452,1986
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intraperitoneal
- DOSE :
- 800 mg/kg
- SEX/DURATION :
- female 10-11 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - musculoskeletal system
- REFERENCE :
- BJEPA5 British Journal of Experimental Pathology. (Blackwell Scientific
Pub. Ltd., POB 88, Oxford, UK) V.1- 1920- Volume(issue)/page/year:
53,5,1972
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intraperitoneal
- DOSE :
- 1800 mg/kg
- SEX/DURATION :
- female 10-12 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - other developmental
abnormalities
- REFERENCE :
- TJADAB Teratology, The International Journal of Abnormal Development. (Alan
R. Liss, Inc., 41 E. 11th St., New York, NY 10003) V.1- 1968-
Volume(issue)/page/year: 6,239,1972
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- DOSE :
- 1 gm/kg
- SEX/DURATION :
- female 11-12 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - musculoskeletal system
- REFERENCE :
- JHMJAX Johns Hopkins Medical Journal. (Baltimore, MD) V.120-151, 1967-82.
Discontinued. Volume(issue)/page/year: 130,105,1972
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- DOSE :
- 1200 mg/kg
- SEX/DURATION :
- female 10-11 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - musculoskeletal system
- REFERENCE :
- TJADAB Teratology, The International Journal of Abnormal Development. (Alan
R. Liss, Inc., 41 E. 11th St., New York, NY 10003) V.1- 1968-
Volume(issue)/page/year: 15,253,1977
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- DOSE :
- 1 gm/kg
- SEX/DURATION :
- female 11 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - other developmental
abnormalities
- REFERENCE :
- TJADAB Teratology, The International Journal of Abnormal Development. (Alan
R. Liss, Inc., 41 E. 11th St., New York, NY 10003) V.1- 1968-
Volume(issue)/page/year: 6,239,1972
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Unreported
- DOSE :
- 500 mg/kg
- SEX/DURATION :
- female 10 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - musculoskeletal system
- REFERENCE :
- TJADAB Teratology, The International Journal of Abnormal Development. (Alan
R. Liss, Inc., 41 E. 11th St., New York, NY 10003) V.1- 1968-
Volume(issue)/page/year: 34,470,1986
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 300 mg/kg
- SEX/DURATION :
- female 9-10 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death,
e.g., stunted fetus)
Reproductive - Specific Developmental Abnormalities - musculoskeletal system
- REFERENCE :
- TJADAB Teratology, The International Journal of Abnormal Development. (Alan
R. Liss, Inc., 41 E. 11th St., New York, NY 10003) V.1- 1968-
Volume(issue)/page/year: 29,427,1984
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 6 gm/kg
- SEX/DURATION :
- female 6-15 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Fertility - pre-implantation mortality (e.g. reduction in
number of implants per female; total number of implants per corpora lutea)
Reproductive - Fertility - post-implantation mortality (e.g. dead and/or
resorbed implants per total number of implants)
Reproductive - Fertility - other measures of fertility
- REFERENCE :
- DCTODJ Drug and Chemical Toxicology. (Marcel Dekker, 270 Madison Ave., New
York, NY 10016) V.1- 1977/78- Volume(issue)/page/year: 3,83,1980
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 6 gm/kg
- SEX/DURATION :
- female 6-15 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death,
e.g., stunted fetus)
Reproductive - Specific Developmental Abnormalities - Central Nervous System
Reproductive - Specific Developmental Abnormalities - craniofacial
(including nose and tongue)
- REFERENCE :
- DCTODJ Drug and Chemical Toxicology. (Marcel Dekker, 270 Madison Ave., New
York, NY 10016) V.1- 1977/78- Volume(issue)/page/year: 3,83,1980
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 6 gm/kg
- SEX/DURATION :
- female 6-15 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - body wall
Reproductive - Specific Developmental Abnormalities - musculoskeletal system
Reproductive - Specific Developmental Abnormalities - urogenital system
- REFERENCE :
- DCTODJ Drug and Chemical Toxicology. (Marcel Dekker, 270 Madison Ave., New
York, NY 10016) V.1- 1977/78- Volume(issue)/page/year: 3,83,1980
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 2 gm/kg
- SEX/DURATION :
- female 8-9 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Fertility - post-implantation mortality (e.g. dead and/or
resorbed implants per total number of implants)
Reproductive - Effects on Embryo or Fetus - fetal death
Reproductive - Specific Developmental Abnormalities - musculoskeletal system
- REFERENCE :
- TJADAB Teratology, The International Journal of Abnormal Development. (Alan
R. Liss, Inc., 41 E. 11th St., New York, NY 10003) V.1- 1968-
Volume(issue)/page/year: 27,33A,1983
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 10 gm/kg
- SEX/DURATION :
- female 8-12 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Newborn - live birth index (measured after birth)
Reproductive - Effects on Newborn - viability index (e.g., # alive at day 4
per # born alive)
Reproductive - Effects on Newborn - growth statistics (e.g.%, reduced weight
gain)
- REFERENCE :
- TCMUD8 Teratogenesis, Carcinogenesis, and Mutagenesis. (Alan R. Liss, Inc.,
41 E. 11th St., New York, NY 10003) V.1- 1980- Volume(issue)/page/year:
6,361,1986
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intraperitoneal
- DOSE :
- 1 gm/kg
- SEX/DURATION :
- female 10 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - musculoskeletal system
- REFERENCE :
- TJADAB Teratology, The International Journal of Abnormal Development. (Alan
R. Liss, Inc., 41 E. 11th St., New York, NY 10003) V.1- 1968-
Volume(issue)/page/year: 11,31,1975
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intraperitoneal
- DOSE :
- 2 gm/kg
- SEX/DURATION :
- female 10 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Fertility - post-implantation mortality (e.g. dead and/or
resorbed implants per total number of implants)
- REFERENCE :
- TJADAB Teratology, The International Journal of Abnormal Development. (Alan
R. Liss, Inc., 41 E. 11th St., New York, NY 10003) V.1- 1968-
Volume(issue)/page/year: 11,31,1975
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intraperitoneal
- DOSE :
- 200 mg/kg
- SEX/DURATION :
- female 8 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - craniofacial
(including nose and tongue)
- REFERENCE :
- TJADAB Teratology, The International Journal of Abnormal Development. (Alan
R. Liss, Inc., 41 E. 11th St., New York, NY 10003) V.1- 1968-
Volume(issue)/page/year: 25(2),28A,1982
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intraperitoneal
- DOSE :
- 1400 mg/kg
- SEX/DURATION :
- female 10 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Fertility - post-implantation mortality (e.g. dead and/or
resorbed implants per total number of implants)
Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death,
e.g., stunted fetus)
Reproductive - Specific Developmental Abnormalities - musculoskeletal system
- REFERENCE :
- DEVPED Development (Cambridge, UK). (Biochemical society Book Depot, POB 32,
Commerce Way, Colchester, Essex CO2 8HP, UK) V.99- 1987-
Volume(issue)/page/year: 107,637,1989
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- DOSE :
- 200 mg/kg
- SEX/DURATION :
- female 8 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - musculoskeletal system
- REFERENCE :
- TJADAB Teratology, The International Journal of Abnormal Development. (Alan
R. Liss, Inc., 41 E. 11th St., New York, NY 10003) V.1- 1968-
Volume(issue)/page/year: 23,26A,1981
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- DOSE :
- 500 mg/kg
- SEX/DURATION :
- female 9 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - musculoskeletal system
- REFERENCE :
- TXAPA9 Toxicology and Applied Pharmacology. (Academic Press, Inc., 1 E.
First St., Duluth, MN 55802) V.1- 1959- Volume(issue)/page/year:
103,238,1990
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Parenteral
- DOSE :
- 1 gm/kg
- SEX/DURATION :
- female 9 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - musculoskeletal system
Reproductive - Effects on Newborn - sex ratio
- REFERENCE :
- TJADAB Teratology, The International Journal of Abnormal Development. (Alan
R. Liss, Inc., 41 E. 11th St., New York, NY 10003) V.1- 1968-
Volume(issue)/page/year: 29(2),35A,1984
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Unreported
- DOSE :
- 4 gm/kg
- SEX/DURATION :
- female 9 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - musculoskeletal system
- REFERENCE :
- TJADAB Teratology, The International Journal of Abnormal Development. (Alan
R. Liss, Inc., 41 E. 11th St., New York, NY 10003) V.1- 1968-
Volume(issue)/page/year: 25(2),49A,1982
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Unreported
- DOSE :
- 2 gm/kg
- SEX/DURATION :
- female 9 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - craniofacial
(including nose and tongue)
- REFERENCE :
- TJADAB Teratology, The International Journal of Abnormal Development. (Alan
R. Liss, Inc., 41 E. 11th St., New York, NY 10003) V.1- 1968-
Volume(issue)/page/year: 29(2),39A,1984
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 1300 mg/kg
- SEX/DURATION :
- female 6-18 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death,
e.g., stunted fetus)
- REFERENCE :
- TJADAB Teratology, The International Journal of Abnormal Development. (Alan
R. Liss, Inc., 41 E. 11th St., New York, NY 10003) V.1- 1968-
Volume(issue)/page/year: 45,629,1992
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 1950 mg/kg
- SEX/DURATION :
- female 6-18 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - musculoskeletal system
- REFERENCE :
- TJADAB Teratology, The International Journal of Abnormal Development. (Alan
R. Liss, Inc., 41 E. 11th St., New York, NY 10003) V.1- 1968-
Volume(issue)/page/year: 45,629,1992
*** REVIEWS ***
TOXICOLOGY REVIEW
JAMAAP JAMA, Journal of the American Medical Association. (AMA, 535 N.
Dearborn St., Chicago, IL 60610) V.1- 1883- Volume(issue)/page/year:
240,251,1978
*** NIOSH STANDARDS DEVELOPMENT AND SURVEILLANCE DATA ***
NIOSH OCCUPATIONAL EXPOSURE SURVEY DATA :
NOES - National Occupational Exposure Survey (1983)
NOES Hazard Code - X4082
No. of Facilities: 193 (estimated)
No. of Industries: 1
No. of Occupations: 5
No. of Employees: 11564 (estimated)
No. of Female Employees: 9953 (estimated)