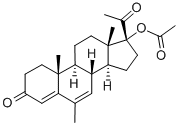
Megestrol acetate cas no:595-33-5
Synonyms: Megace ES;Pregna-4,6-diene-3,20-dione, 17-(acetyloxy)-6-methyl-;Ovarid;Megace;Ovaban (Veterinary);Megeron;6-Methyl-delta4,6-pregnadien-17alpha-ol-3,20-dione acetate;6-Methyl-6-dehydro-17.alpha.-acetoxyprogesterone;Megace (TN);SC 10363;Megestryl acetate;Pregna-4,6-diene-3, 20-dione, 17-hydroxy-6-methyl-, acetate;Pallace;Ovaban;S4;6-Methyl-17-alpha-hydroxy-delta(sup 6)-progesterone acetate;SC-10363;SC10363;Niagestin;Progesterone, 6-dehydro-17-hydroxy-6-methyl-, acetate;6-Methyl-delta(sup 4,6)-pregnadien-17-alpha-ol-3,20-dione acetate;Megestrol acetate [USAN];17alpha-Acetoxy-6-dehydro-6-methylprogesterone;Megestrol acetate (USAN);Megestat;6-Dehydro-6-methyl-17.alpha.-acetoxyprogesterone;17-Hydroxy-6-methylpregna-4,6-diene-3,20-dione acetate;Megestrol acetole [Progestins];6-MethleneProgesterone Acetate;Pregna-4,6-diene-3, 20-dione, 17- (acetyloxy)-6-methyl-;BDH 1298;
NameMegestrol acetate
CAS595-33-5
SynonymsMegace ES;Pregna-4,6-diene-3,20-dione, 17-(acetyloxy)-6-methyl-;Ovarid;Megace;Ovaban (Veterinary);Megeron;6-Methyl-delta4,6-pregnadien-17alpha-ol-3,20-dione acetate;6-Methyl-6-dehydro-17.alpha.-acetoxyprogesterone;Megace (TN);SC 10363;Megestryl acetate;Pregna-4,6-diene-3, 20-dione, 17-hydroxy-6-methyl-, acetate;Pallace;Ovaban;S4;6-Methyl-17-alpha-hydroxy-delta(sup 6)-progesterone acetate;SC-10363;SC10363;Niagestin;Progesterone, 6-dehydro-17-hydroxy-6-methyl-, acetate;6-Methyl-delta(sup 4,6)-pregnadien-17-alpha-ol-3,20-dione acetate;Megestrol acetate [USAN];17alpha-Acetoxy-6-dehydro-6-methylprogesterone;Megestrol acetate (USAN);Megestat;6-Dehydro-6-methyl-17.alpha.-acetoxyprogesterone;17-Hydroxy-6-methylpregna-4,6-diene-3,20-dione acetate;Megestrol acetole [Progestins];6-MethleneProgesterone Acetate;Pregna-4,6-diene-3, 20-dione, 17- (acetyloxy)-6-methyl-;BDH 1298;
EINECS(EC#)209-864-5
Molecular FormulaC24H32O4
Molecular Weight384.51
AppearanceCrystalline Solid
refractive index11 ° (C=2, CHCl3)
storage tempRefrigerator
Hazard Xn:Harmful
Risk R40;R48
Safety Hazard Codes:Xn
Risk Statements:40-48
40:Limited evidence of a carcinogenic effect
48:Danger of serious damage to health by prolonged exposure
Safety Statements:22-24/25
22:Do not breathe dust
24/25:Avoid contact with skin and eyes
WGK Germany:3
CHEMICAL IDENTIFICATION
- RTECS NUMBER :
- TU4075000
- CHEMICAL NAME :
- Pregna-4,6-diene-3,20-dione, 17-hydroxy-6-methyl-,
acetate
- CAS REGISTRY NUMBER :
- 595-33-5
- LAST UPDATED :
- 199806
- DATA ITEMS CITED :
- 40
- MOLECULAR FORMULA :
- C24-H32-O4
- MOLECULAR WEIGHT :
- 384.56
- WISWESSER LINE NOTATION :
- L E5 B666 OV KU MUTJ A1 E1 FV1 FOV1 L1
HEALTH HAZARD DATA
ACUTE TOXICITY DATA
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 56 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Mammal - dog
- DOSE/DURATION :
- 256 mg/kg/7Y-C
- TOXIC EFFECTS :
- Tumorigenic - Carcinogenic by RTECS criteria
Skin and Appendages - tumors
Endocrine - diabetes mellitus
- TYPE OF TEST :
- TD - Toxic dose (other than lowest)
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Mammal - dog
- DOSE/DURATION :
- 182 mg/kg/2Y-C
- TOXIC EFFECTS :
- Tumorigenic - equivocal tumorigenic agent by RTECS criteria
Skin and Appendages - tumors
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 210 ug/kg
- SEX/DURATION :
- female 21 day(s) pre-mating
- TOXIC EFFECTS :
- Reproductive - Fertility - female fertility index (e.g. # females pregnant
per # sperm positive females; # females pregnant per # females mated)
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 1825 ug/kg
- SEX/DURATION :
- female 52 week(s) pre-mating
- TOXIC EFFECTS :
- Reproductive - Fertility - female fertility index (e.g. # females pregnant
per # sperm positive females; # females pregnant per # females mated)
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 2430 ug/kg
- SEX/DURATION :
- female 34 week(s) pre-mating
- TOXIC EFFECTS :
- Reproductive - Maternal Effects - menstrual cycle changes or disorders
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 1820 ug/kg
- SEX/DURATION :
- female 26 week(s) pre-mating
- TOXIC EFFECTS :
- Reproductive - Maternal Effects - ovaries, fallopian tubes
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Implant
- DOSE :
- 1800 ug/kg
- SEX/DURATION :
- female 30 day(s) pre-mating
- TOXIC EFFECTS :
- Reproductive - Fertility - female fertility index (e.g. # females pregnant
per # sperm positive females; # females pregnant per # females mated)
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Implant
- DOSE :
- 1440 ug/kg
- SEX/DURATION :
- female 43 week(s) pre-mating
- TOXIC EFFECTS :
- Reproductive - Fertility - female fertility index (e.g. # females pregnant
per # sperm positive females; # females pregnant per # females mated)
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Implant
- DOSE :
- 1440 ug/kg
- SEX/DURATION :
- female 40 week(s) pre-mating
- TOXIC EFFECTS :
- Reproductive - Maternal Effects - menstrual cycle changes or disorders
Reproductive - Fertility - female fertility index (e.g. # females pregnant
per # sperm positive females; # females pregnant per # females mated)
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Implant
- DOSE :
- 1840 ug/kg
- SEX/DURATION :
- female 43 week(s) pre-mating
- TOXIC EFFECTS :
- Reproductive - Maternal Effects - menstrual cycle changes or disorders
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 1200 mg/kg
- SEX/DURATION :
- male 30 day(s) pre-mating
- TOXIC EFFECTS :
- Reproductive - Paternal Effects - testes, epididymis, sperm duct
Reproductive - Paternal Effects - prostate, seminal vesicle, Cowper's gland,
accessory glands
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 2790 ug/kg
- SEX/DURATION :
- female 50 week(s) pre-mating
female 1-22 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Fertility - female fertility index (e.g. # females pregnant
per # sperm positive females; # females pregnant per # females mated)
Reproductive - Fertility - pre-implantation mortality (e.g. reduction in
number of implants per female; total number of implants per corpora lutea)
Reproductive - Fertility - post-implantation mortality (e.g. dead and/or
resorbed implants per total number of implants)
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 9 mg/kg
- SEX/DURATION :
- female 15-20 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - urogenital system
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 15 mg/kg
- SEX/DURATION :
- male 15 day(s) pre-mating
- TOXIC EFFECTS :
- Reproductive - Paternal Effects - prostate, seminal vesicle, Cowper's gland,
accessory glands
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 15 mg/kg
- SEX/DURATION :
- female 15 day(s) pre-mating
- TOXIC EFFECTS :
- Reproductive - Maternal Effects - uterus, cervix, vagina
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- DOSE :
- 140 mg/kg
- SEX/DURATION :
- female 1-7 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Fertility - litter size (e.g. # fetuses per litter; measured
before birth)
Reproductive - Fertility - other measures of fertility
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- DOSE :
- 7 mg/kg
- SEX/DURATION :
- female 14 day(s) pre-mating
- TOXIC EFFECTS :
- Reproductive - Maternal Effects - ovaries, fallopian tubes
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- DOSE :
- 70 mg/kg
- SEX/DURATION :
- female 14 day(s) pre-mating
- TOXIC EFFECTS :
- Reproductive - Maternal Effects - uterus, cervix, vagina
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- DOSE :
- 14 mg/kg
- SEX/DURATION :
- female 14 day(s) pre-mating
- TOXIC EFFECTS :
- Reproductive - Fertility - other measures of fertility
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Unreported
- DOSE :
- 15 mg/kg
- SEX/DURATION :
- female 30 day(s) pre-mating
- TOXIC EFFECTS :
- Reproductive - Maternal Effects - uterus, cervix, vagina
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 3 mg/kg
- SEX/DURATION :
- female 3 day(s) pre-mating
- TOXIC EFFECTS :
- Reproductive - Maternal Effects - uterus, cervix, vagina
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 16800 ug/kg
- SEX/DURATION :
- female 21 day(s) pre-mating
- TOXIC EFFECTS :
- Reproductive - Fertility - female fertility index (e.g. # females pregnant
per # sperm positive females; # females pregnant per # females mated)
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 4600 ug/kg
- SEX/DURATION :
- female 23 day(s) pre-mating
- TOXIC EFFECTS :
- Reproductive - Maternal Effects - uterus, cervix, vagina
Reproductive - Maternal Effects - other effects
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- DOSE :
- 100 ug/kg
- SEX/DURATION :
- female 5 day(s) pre-mating
- TOXIC EFFECTS :
- Reproductive - Fertility - other measures of fertility
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 2 mg/kg
- SEX/DURATION :
- female 1 day(s) pre-mating
- TOXIC EFFECTS :
- Reproductive - Fertility - female fertility index (e.g. # females pregnant
per # sperm positive females; # females pregnant per # females mated)
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 625 ug/kg
- SEX/DURATION :
- female 1 day(s) pre-mating
- TOXIC EFFECTS :
- Reproductive - Fertility - mating performance (e.g. # sperm positive females
per # females mated; # copulations per # estrus cycles)
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 150 ug/kg
- SEX/DURATION :
- female 3 day(s) pre-mating
- TOXIC EFFECTS :
- Reproductive - Fertility - female fertility index (e.g. # females pregnant
per # sperm positive females; # females pregnant per # females mated)
Reproductive - Fertility - other measures of fertility
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 37500 ng/kg
- SEX/DURATION :
- female 3 day(s) pre-mating
- TOXIC EFFECTS :
- Reproductive - Fertility - litter size (e.g. # fetuses per litter; measured
before birth)
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 8 ug/kg
- SEX/DURATION :
- female 1 day(s) pre-mating
- TOXIC EFFECTS :
- Reproductive - Fertility - other measures of fertility
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- DOSE :
- 50 ug/kg
- SEX/DURATION :
- female 5 day(s) pre-mating
- TOXIC EFFECTS :
- Reproductive - Maternal Effects - uterus, cervix, vagina
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intramuscular
- DOSE :
- 128 ug/kg
- SEX/DURATION :
- female 16 day(s) pre-mating
- TOXIC EFFECTS :
- Reproductive - Maternal Effects - menstrual cycle changes or disorders
- TYPE OF TEST :
- DNA adduct
- TYPE OF TEST :
- Unscheduled DNA synthesis
MUTATION DATA
- TEST SYSTEM :
- Rodent - mouse
- DOSE/DURATION :
- 200 mg/kg
- REFERENCE :
- JOENAK Journal of Endocrinology. (Biochemical Soc. Book Depot, POB 32,
Commerce Way, Colchester, Essex CO2 8HP, UK) V.1- 1939-
Volume(issue)/page/year: 60,167,1974
*** REVIEWS ***
IARC Cancer Review:Animal Limited Evidence
IMEMDT IARC Monographs on the Evaluation of Carcinogenic Risk of Chemicals
to Man. (WHO Publications Centre USA, 49 Sheridan Ave., Albany, NY 12210)
V.1- 1972- Volume(issue)/page/year: 21,431,1979
TOXICOLOGY REVIEW
CMROCX Current Medical Research and Opinion. (Clayton-Wray Pub. Ltd., 1a
High St., Alton, Hants., UK) V.1- 1972- Volume(issue)/page/year:
4,309,1976
*** NIOSH STANDARDS DEVELOPMENT AND SURVEILLANCE DATA ***
NIOSH OCCUPATIONAL EXPOSURE SURVEY DATA :
NOES - National Occupational Exposure Survey (1983)
NOES Hazard Code - X6284
No. of Facilities: 22 (estimated)
No. of Industries: 1
No. of Occupations: 1
No. of Employees: 771 (estimated)
No. of Female Employees: 372 (estimated)