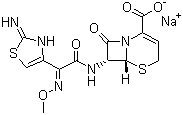
5-Thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid,7-[[(2Z)-(2-amino-4-thiazolyl)(methoxyimino)acetyl]amino]-8-oxo-, monosodiumsalt, (6R,7R)- cas no:68401-82-1
Synonyms: (6R,7R)-7-{[(2Z)-2-(2-Amino-1,3-thiazol-4-yl)-2-(méthoxyimino)acétyl]amino}-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ène-2-carboxylate de sodium;5-Thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid, 7-[[(2Z)-2-(2-amino-4-thiazolyl)-2-(methoxyimino)-1-oxoethyl]amino]-8-oxo-, sodium salt, (6R,7R)- (1:1);Natrium-(6R,7R)-7-{[(2Z)-2-(2-amino-1,3-thiazol-4-yl)-2-(methoxyimino)acetyl]amino}-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-en-2-carboxylat;Cefizox in plastic container;
Name5-Thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid,7-[[(2Z)-(2-amino-4-thiazolyl)(methoxyimino)acetyl]amino]-8-oxo-, monosodiumsalt, (6R,7R)-
CAS68401-82-1
Synonyms(6R,7R)-7-{[(2Z)-2-(2-Amino-1,3-thiazol-4-yl)-2-(méthoxyimino)acétyl]amino}-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ène-2-carboxylate de sodium;5-Thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid, 7-[[(2Z)-2-(2-amino-4-thiazolyl)-2-(methoxyimino)-1-oxoethyl]amino]-8-oxo-, sodium salt, (6R,7R)- (1:1);Natrium-(6R,7R)-7-{[(2Z)-2-(2-amino-1,3-thiazol-4-yl)-2-(methoxyimino)acetyl]amino}-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-en-2-carboxylat;Cefizox in plastic container;
Molecular FormulaC13H12N5NaO5S2
Molecular Weight405.38
AppearanceWhite to light yellow crystalline powder
Safety Mildly toxic by intravenous and intraperitoneal routes. Experimental teratogenic and reproductive effects. When heated to decomposition it emits toxic fumes of SOx, NOx, and Na2O.
CHEMICAL IDENTIFICATION
- RTECS NUMBER :
- XI0368000
- CHEMICAL NAME :
- 5-Thia-1-azabicyclo(4.2.0)oct-2-ene-2-carboxylic
acid, 7-(((2-amino-4-thiazolyl)-
(methoxyimino)acetyl)amino)-8-oxo-, monosodium salt,
(6R-(6-alpha,7-beta(Z)))-
- CAS REGISTRY NUMBER :
- 68401-82-1
- LAST UPDATED :
- 199306
- DATA ITEMS CITED :
- 18
- MOLECULAR FORMULA :
- C13-H12-N5-O5-S2.Na
- MOLECULAR WEIGHT :
- 405.41
- WISWESSER LINE NOTATION :
- T46 ANV ES GUTJ GVO CMVYUNO1&- ET5N CSJ BZ &-NA-
HEALTH HAZARD DATA
ACUTE TOXICITY DATA
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- >10 gm/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- ARZNAD Arzneimittel-Forschung. Drug Research. (Editio Cantor Verlag,
Postfach 1255, W-7960 Aulendorf, Fed. Rep. Ger.) V.1- 1951-
Volume(issue)/page/year: 30,1669,1980
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intraperitoneal
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 8130 mg/kg
- TOXIC EFFECTS :
- Peripheral Nerve and Sensation - flaccid paralysis without anesthesia
(usually neuromuscular blockage)
Lungs, Thorax, or Respiration - other changes
- REFERENCE :
- ARZNAD Arzneimittel-Forschung. Drug Research. (Editio Cantor Verlag,
Postfach 1255, W-7960 Aulendorf, Fed. Rep. Ger.) V.1- 1951-
Volume(issue)/page/year: 30,1669,1980
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- >10 gm/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- ARZNAD Arzneimittel-Forschung. Drug Research. (Editio Cantor Verlag,
Postfach 1255, W-7960 Aulendorf, Fed. Rep. Ger.) V.1- 1951-
Volume(issue)/page/year: 30,1669,1980
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 5570 mg/kg
- TOXIC EFFECTS :
- Peripheral Nerve and Sensation - flaccid paralysis without anesthesia
(usually neuromuscular blockage)
Lungs, Thorax, or Respiration - other changes
- REFERENCE :
- ARZNAD Arzneimittel-Forschung. Drug Research. (Editio Cantor Verlag,
Postfach 1255, W-7960 Aulendorf, Fed. Rep. Ger.) V.1- 1951-
Volume(issue)/page/year: 30,1669,1980
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- >10 gm/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- ARZNAD Arzneimittel-Forschung. Drug Research. (Editio Cantor Verlag,
Postfach 1255, W-7960 Aulendorf, Fed. Rep. Ger.) V.1- 1951-
Volume(issue)/page/year: 30,1669,1980
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intraperitoneal
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 8930 mg/kg
- TOXIC EFFECTS :
- Peripheral Nerve and Sensation - flaccid paralysis without anesthesia
(usually neuromuscular blockage)
Lungs, Thorax, or Respiration - other changes
- REFERENCE :
- ARZNAD Arzneimittel-Forschung. Drug Research. (Editio Cantor Verlag,
Postfach 1255, W-7960 Aulendorf, Fed. Rep. Ger.) V.1- 1951-
Volume(issue)/page/year: 30,1669,1980
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- >10 gm/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- ARZNAD Arzneimittel-Forschung. Drug Research. (Editio Cantor Verlag,
Postfach 1255, W-7960 Aulendorf, Fed. Rep. Ger.) V.1- 1951-
Volume(issue)/page/year: 30,1669,1980
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 5150 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- YAKUD5 Gekkan Yakuji. Pharmaceuticals Monthly. (Yakugyo Jihosha, Inaoka
Bldg., 2-36 Jinbo-cho, Kanda, Chiyoda-ku, Tokyo 101, Japan) V.1- 1959-
Volume(issue)/page/year: 24,905,1982
- TYPE OF TEST :
- LDLo - Lowest published lethal dose
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Mammal - dog
- DOSE/DURATION :
- >3200 mg/kg
- TOXIC EFFECTS :
- Behavioral - somnolence (general depressed activity)
Cardiac - cardiomyopathy including infarction
- REFERENCE :
- ARZNAD Arzneimittel-Forschung. Drug Research. (Editio Cantor Verlag,
Postfach 1255, W-7960 Aulendorf, Fed. Rep. Ger.) V.1- 1951-
Volume(issue)/page/year: 30,1669,1980
- TYPE OF TEST :
- LDLo - Lowest published lethal dose
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rabbit
- DOSE/DURATION :
- >3200 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- ARZNAD Arzneimittel-Forschung. Drug Research. (Editio Cantor Verlag,
Postfach 1255, W-7960 Aulendorf, Fed. Rep. Ger.) V.1- 1951-
Volume(issue)/page/year: 30,1669,1980
- TYPE OF TEST :
- LDLo - Lowest published lethal dose
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Rodent - rabbit
- DOSE/DURATION :
- >3200 mg/kg
- TOXIC EFFECTS :
- Behavioral - muscle weakness
Gastrointestinal - hypermotility, diarrhea
Nutritional and Gross Metabolic - weight loss or decreased weight gain
- REFERENCE :
- ARZNAD Arzneimittel-Forschung. Drug Research. (Editio Cantor Verlag,
Postfach 1255, W-7960 Aulendorf, Fed. Rep. Ger.) V.1- 1951-
Volume(issue)/page/year: 30,1669,1980
** OTHER MULTIPLE DOSE TOXICITY DATA **
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 29120 mg/kg/13W-I
- TOXIC EFFECTS :
- Blood - hemorrhage
Blood - changes in serum composition (e.g. TP, bilirubin, cholesterol)
Skin and Appendages - dermatitis, other (after systemic exposure)
- REFERENCE :
- ARZNAD Arzneimittel-Forschung. Drug Research. (Editio Cantor Verlag,
Postfach 1255, W-7960 Aulendorf, Fed. Rep. Ger.) V.1- 1951-
Volume(issue)/page/year: 30,1669,1980
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 58240 mg/kg/26W-I
- TOXIC EFFECTS :
- Blood - hemorrhage
Blood - changes in serum composition (e.g. TP, bilirubin, cholesterol)
Skin and Appendages - dermatitis, other (after systemic exposure)
- REFERENCE :
- ARZNAD Arzneimittel-Forschung. Drug Research. (Editio Cantor Verlag,
Postfach 1255, W-7960 Aulendorf, Fed. Rep. Ger.) V.1- 1951-
Volume(issue)/page/year: 30,1669,1980
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Mammal - dog
- DOSE/DURATION :
- 58240 mg/kg/26W-I
- TOXIC EFFECTS :
- Blood - hemorrhage
Skin and Appendages - dermatitis, other (after systemic exposure)
Biochemical - Enzyme inhibition, induction, or change in blood or tissue
levels - transaminases
- REFERENCE :
- ARZNAD Arzneimittel-Forschung. Drug Research. (Editio Cantor Verlag,
Postfach 1255, W-7960 Aulendorf, Fed. Rep. Ger.) V.1- 1951-
Volume(issue)/page/year: 30,1669,1980
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intramuscular
- SPECIES OBSERVED :
- Mammal - dog
- DOSE/DURATION :
- 8960 mg/kg/4W-I
- TOXIC EFFECTS :
- Blood - hemorrhage
Skin and Appendages - dermatitis, other (after systemic exposure)
Biochemical - Enzyme inhibition, induction, or change in blood or tissue
levels - phosphokinase
- REFERENCE :
- ARZNAD Arzneimittel-Forschung. Drug Research. (Editio Cantor Verlag,
Postfach 1255, W-7960 Aulendorf, Fed. Rep. Ger.) V.1- 1951-
Volume(issue)/page/year: 30,1669,1980
** REPRODUCTIVE DATA **
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- DOSE :
- 10 gm/kg
- SEX/DURATION :
- male 9 week(s) pre-mating
female 2 week(s) pre-mating - 22 day(s) after
conception
- TOXIC EFFECTS :
- Reproductive - Effects on Newborn - growth statistics (e.g.%, reduced weight
gain)
- REFERENCE :
- ARZNAD Arzneimittel-Forschung. Drug Research. (Editio Cantor Verlag,
Postfach 1255, W-7960 Aulendorf, Fed. Rep. Ger.) V.1- 1951-
Volume(issue)/page/year: 30,1669,1980
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- DOSE :
- 27 gm/kg
- SEX/DURATION :
- male 9 week(s) pre-mating
female 2 week(s) pre-mating - 7 day(s) after
conception
- TOXIC EFFECTS :
- Reproductive - Fertility - post-implantation mortality (e.g. dead and/or
resorbed implants per total number of implants)
Reproductive - Fertility - litter size (e.g. # fetuses per litter; measured
before birth)
- REFERENCE :
- JJANAX Japanese Journal of Antibiotics. (Japan Antibiotics Research Assoc.,
2-20-8 Kamiosaki, Shinagawa-ku, Tokyo 141, Japan) V.21- 1968-
Volume(issue)/page/year: 34,466,1981
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- DOSE :
- 1100 mg/kg
- SEX/DURATION :
- female 7-17 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - musculoskeletal system
- REFERENCE :
- JJANAX Japanese Journal of Antibiotics. (Japan Antibiotics Research Assoc.,
2-20-8 Kamiosaki, Shinagawa-ku, Tokyo 141, Japan) V.21- 1968-
Volume(issue)/page/year: 34,466,1981