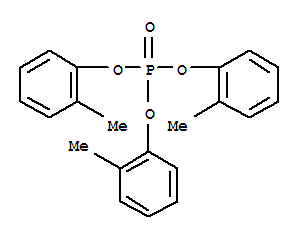
Hazard Toxic by ingestion and skin absorption. The o- isomer is highly toxic. TLV: 0.1 mg/m3 (skin); Not Classifiable as a Human Carcinogen.
Risk R51/53
Safety Poison by subcutaneous, intramuscular, intravenous, and intraperitoneal routes. Moderately toxic by ingestion. Most of the cases of tri-o-cresyl phosphate poisoning have followed its ingestion. In 1930, some 15,000 persons were affected in the United States, and of these, 10 died. The responsible material was found to be an alcoholic drink known as Jamaica ginger, or “jake.” This beverage had been adulterated with about 2% of tri-o-cresyl phosphate. The affected persons developed a polyneuritis, which progressed, in many cases, with degeneration of the peripheral motor nerves, the anterior horn cells, and the pyramidal tracts. Sensory changes were absent. Since 1930 there have been several other outbreaks of poisoning following ingestion of the material. Tri-o-cresyl phosphate is more toxic than the m-form, and much more so than tri-p-cresyl phosphate or triphenyl phosphate. Experimental reproductive effects.Combustible when exposed to heat or flame. Can react with oxidizing materials. To fight fire, use CO2, dry chemical. When heated to decomposition it emits highly toxic fumes of POx. See also PHOSPHATES.Analytical Methods: For occupational chemical analysis use NIOSH: Triorthocresyl Phosphate, S209.