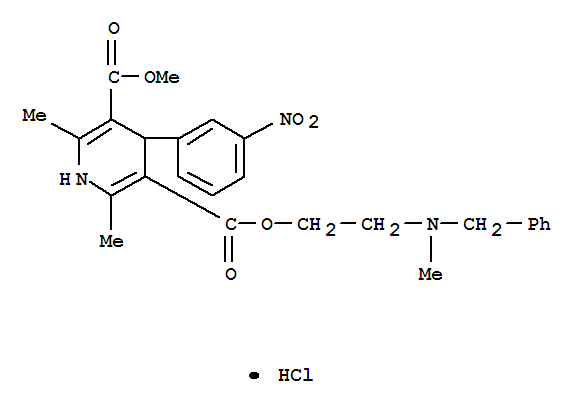
3,5-Pyridinedicarboxylicacid, 1,4-dihydro-2,6-dimethyl-4-(3-nitrophenyl)-, 3-methyl5-[2-[methyl(phenylmethyl)amino]ethyl] ester, hydrochloride (1:1) cas no:54527-84-3
Synonyms: 3,5-Pyridinedicarboxylicacid, 1,4-dihydro-2,6-dimethyl-4-(3-nitrophenyl)-, methyl2-[methyl(phenylmethyl)amino]ethyl ester, monohydrochloride (9CI);2,6-Dimethyl-4-(3-nitrophenyl)-1,4-dihydropyridine-3,5-dicarboxylic acid3-[2-(N-benzyl-N-methylamino)]-ethyl ester 5-methyl ester hydrochloride;Barizin;Bionicard;Cardene;Cardene (pharmaceutical);Lecibral;Nerdipina;Nicapress;Nicardal;Nimicor;Perdipina;
Name3,5-Pyridinedicarboxylicacid, 1,4-dihydro-2,6-dimethyl-4-(3-nitrophenyl)-, 3-methyl5-[2-[methyl(phenylmethyl)amino]ethyl] ester, hydrochloride (1:1)
CAS54527-84-3
Synonyms3,5-Pyridinedicarboxylicacid, 1,4-dihydro-2,6-dimethyl-4-(3-nitrophenyl)-, methyl2-[methyl(phenylmethyl)amino]ethyl ester, monohydrochloride (9CI);2,6-Dimethyl-4-(3-nitrophenyl)-1,4-dihydropyridine-3,5-dicarboxylic acid3-[2-(N-benzyl-N-methylamino)]-ethyl ester 5-methyl ester hydrochloride;Barizin;Bionicard;Cardene;Cardene (pharmaceutical);Lecibral;Nerdipina;Nicapress;Nicardal;Nimicor;Perdipina;
EINECS(EC#)259-198-4
Molecular FormulaC26H30ClN3O6
Molecular Weight516.04
AppearanceYellow solid
storage temp2-8°C
Globally Harmonized System of Classification adn Labelling of Chemicals(GHS)
Hazardclass6.1(b)
Hazard T: Toxic;
Risk R23/24/25
Safety Poison by ingestion, intravenous, and intraperitoneal routes. Moderately toxic by subcutaneous route. Human systemic effects: decreased urine volume or anuria. Experimental reproductive effects. When heated to decomposition it emits toxic fumes of NOx and HCl.
CHEMICAL IDENTIFICATION
- RTECS NUMBER :
- US7972100
- CHEMICAL NAME :
- 3,5-Pyridinedicarboxylic acid,
1,4-dihydro-2,6-dimethyl-4-(m-nitrophenyl)-,
2-(benzylmethyl amino)ethyl methyl ester,
monohydrochloride
- CAS REGISTRY NUMBER :
- 54527-84-3
- LAST UPDATED :
- 199703
- DATA ITEMS CITED :
- 25
- MOLECULAR FORMULA :
- C26-H29-N3-O6.Cl-H
- MOLECULAR WEIGHT :
- 516.04
- WISWESSER LINE NOTATION :
- T6M DHJ B1 CVO1 DR CNW& EVO2N1&1R& F1 &GH
HEALTH HAZARD DATA
ACUTE TOXICITY DATA
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Human - man
- DOSE/DURATION :
- 26 mg/kg/30D-I
- TOXIC EFFECTS :
- Kidney, Ureter, Bladder - urine volume decreased
Kidney, Ureter, Bladder - other changes
- REFERENCE :
- JAMAAP JAMA, Journal of the American Medical Association. (AMA, 535 N.
Dearborn St., Chicago, IL 60610) V.1- 1883- Volume(issue)/page/year:
258,3388,1987
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Human - woman
- DOSE/DURATION :
- 1800 ug/kg/36H-I
- TOXIC EFFECTS :
- Kidney, Ureter, Bladder - urine volume decreased
- REFERENCE :
- JAMAAP JAMA, Journal of the American Medical Association. (AMA, 535 N.
Dearborn St., Chicago, IL 60610) V.1- 1883- Volume(issue)/page/year:
258,3388,1987
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 184 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- USXXAM United States Patent Document. (U.S. Patent Office, Box 9,
Washington, DC 20231) Volume(issue)/page/year: #4565823
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intraperitoneal
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 155 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- NIIRDN Drugs in Japan (Ethical Drugs). (Yakugyo Jiho Co., Ltd., Tokyo,
Japan) Volume(issue)/page/year: 6,543,1982
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 606 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- USXXAM United States Patent Document. (U.S. Patent Office, Box 9,
Washington, DC 20231) Volume(issue)/page/year: #4565823
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 15500 ug/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- ARZNAD Arzneimittel-Forschung. Drug Research. (Editio Cantor Verlag,
Postfach 1255, W-7960 Aulendorf, Fed. Rep. Ger.) V.1- 1951-
Volume(issue)/page/year: 26,2172,1976
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 322 mg/kg
- TOXIC EFFECTS :
- Behavioral - tremor
Behavioral - convulsions or effect on seizure threshold
- REFERENCE :
- CYLPDN Zhongguo Yaoli Xuebao. Acta Pharmacologica Sinica. Chinese Journal
of Pharmacology. (China International Book Trading Corp., POB 2820,
Beijing, Peop. Rep. China) V.1- 1980- Volume(issue)/page/year:
4,97,1983
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intraperitoneal
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 123 mg/kg
- TOXIC EFFECTS :
- Behavioral - tremor
Behavioral - convulsions or effect on seizure threshold
- REFERENCE :
- CYLPDN Zhongguo Yaoli Xuebao. Acta Pharmacologica Sinica. Chinese Journal
of Pharmacology. (China International Book Trading Corp., POB 2820,
Beijing, Peop. Rep. China) V.1- 1980- Volume(issue)/page/year:
4,97,1983
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 540 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- NIIRDN Drugs in Japan (Ethical Drugs). (Yakugyo Jiho Co., Ltd., Tokyo,
Japan) Volume(issue)/page/year: 6,543,1982
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 19900 ug/kg
- TOXIC EFFECTS :
- Sense Organs and Special Senses (Eye) - ptosis
Behavioral - ataxia
Lungs, Thorax, or Respiration - dyspnea
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 18,301,1979
- TYPE OF TEST :
- LD - Lethal dose
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Mammal - dog
- DOSE/DURATION :
- >60 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- USXXAM United States Patent Document. (U.S. Patent Office, Box 9,
Washington, DC 20231) Volume(issue)/page/year: #4565823
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Mammal - dog
- DOSE/DURATION :
- 5 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- USXXAM United States Patent Document. (U.S. Patent Office, Box 9,
Washington, DC 20231) Volume(issue)/page/year: #4565823
** OTHER MULTIPLE DOSE TOXICITY DATA **
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 18200 mg/kg/26W-I
- TOXIC EFFECTS :
- Liver - fatty liver degeneration
Blood - changes in serum composition (e.g. TP, bilirubin, cholesterol)
Biochemical - Enzyme inhibition, induction, or change in blood or tissue
levels - transaminases
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 18,391,1979
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Unreported
- SPECIES OBSERVED :
- Mammal - dog
- DOSE/DURATION :
- 4550 mg/kg/26W-I
- TOXIC EFFECTS :
- Behavioral - fluid intake
Cardiac - changes in heart weight
Liver - changes in liver weight
- REFERENCE :
- TOLED5 Toxicology Letters. (Elsevier Science Pub. B.V., POB 211, 1000 AE
Amsterdam, Netherlands) V.1- 1977- Volume(issue)/page/year: 4,57,1979
** REPRODUCTIVE DATA **
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 1050 mg/kg
- SEX/DURATION :
- female 14 day(s) pre-mating
female 1-7 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Fertility - litter size (e.g. # fetuses per litter; measured
before birth)
- REFERENCE :
- KSRNAM Kiso to Rinsho. Clinical Report. (Yubunsha Co., Ltd., 1-5, Kanda
Suda-Cho, Chiyoda-ku, KS Bldg., Tokyo 101, Japan) V.1- 1960-
Volume(issue)/page/year: 13,1160,1979
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 2100 mg/kg
- SEX/DURATION :
- female 14 day(s) pre-mating
female 1-7 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Fertility - pre-implantation mortality (e.g. reduction in
number of implants per female; total number of implants per corpora lutea)
- REFERENCE :
- KSRNAM Kiso to Rinsho. Clinical Report. (Yubunsha Co., Ltd., 1-5, Kanda
Suda-Cho, Chiyoda-ku, KS Bldg., Tokyo 101, Japan) V.1- 1960-
Volume(issue)/page/year: 13,1160,1979
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 2700 mg/kg
- SEX/DURATION :
- female 17-22 day(s) after conception
lactating female 21 day(s) post-birth
- TOXIC EFFECTS :
- Reproductive - Effects on Newborn - viability index (e.g., # alive at day 4
per # born alive)
- REFERENCE :
- KSRNAM Kiso to Rinsho. Clinical Report. (Yubunsha Co., Ltd., 1-5, Kanda
Suda-Cho, Chiyoda-ku, KS Bldg., Tokyo 101, Japan) V.1- 1960-
Volume(issue)/page/year: 13,1160,1979
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 1350 mg/kg
- SEX/DURATION :
- female 17-22 day(s) after conception
lactating female 21 day(s) post-birth
- TOXIC EFFECTS :
- Reproductive - Effects on Newborn - growth statistics (e.g.%, reduced weight
gain)
- REFERENCE :
- KSRNAM Kiso to Rinsho. Clinical Report. (Yubunsha Co., Ltd., 1-5, Kanda
Suda-Cho, Chiyoda-ku, KS Bldg., Tokyo 101, Japan) V.1- 1960-
Volume(issue)/page/year: 13,1160,1979
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intraperitoneal
- DOSE :
- 3360 mg/kg
- SEX/DURATION :
- male 9 week(s) pre-mating
female 2 week(s) pre-mating - 1 week(s) after
conception
- TOXIC EFFECTS :
- Reproductive - Fertility - pre-implantation mortality (e.g. reduction in
number of implants per female; total number of implants per corpora lutea)
Reproductive - Fertility - post-implantation mortality (e.g. dead and/or
resorbed implants per total number of implants)
- REFERENCE :
- KSRNAM Kiso to Rinsho. Clinical Report. (Yubunsha Co., Ltd., 1-5, Kanda
Suda-Cho, Chiyoda-ku, KS Bldg., Tokyo 101, Japan) V.1- 1960-
Volume(issue)/page/year: 22,1411,1988
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravenous
- DOSE :
- 110 mg/kg
- SEX/DURATION :
- female 7-17 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Fertility - litter size (e.g. # fetuses per litter; measured
before birth)
Reproductive - Effects on Embryo or Fetus - fetal death
Reproductive - Effects on Newborn - viability index (e.g., # alive at day 4
per # born alive)
- REFERENCE :
- KSRNAM Kiso to Rinsho. Clinical Report. (Yubunsha Co., Ltd., 1-5, Kanda
Suda-Cho, Chiyoda-ku, KS Bldg., Tokyo 101, Japan) V.1- 1960-
Volume(issue)/page/year: 20,5419,1986
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravenous
- DOSE :
- 55 mg/kg
- SEX/DURATION :
- female 7-17 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Newborn - live birth index (measured after birth)
- REFERENCE :
- KSRNAM Kiso to Rinsho. Clinical Report. (Yubunsha Co., Ltd., 1-5, Kanda
Suda-Cho, Chiyoda-ku, KS Bldg., Tokyo 101, Japan) V.1- 1960-
Volume(issue)/page/year: 20,5419,1986
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravenous
- DOSE :
- 30 mg/kg
- SEX/DURATION :
- female 17-22 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Newborn - stillbirth
- REFERENCE :
- KSRNAM Kiso to Rinsho. Clinical Report. (Yubunsha Co., Ltd., 1-5, Kanda
Suda-Cho, Chiyoda-ku, KS Bldg., Tokyo 101, Japan) V.1- 1960-
Volume(issue)/page/year: 22,1419,1988
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravenous
- DOSE :
- 60 mg/kg
- SEX/DURATION :
- female 17-22 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death,
e.g., stunted fetus)
- REFERENCE :
- KSRNAM Kiso to Rinsho. Clinical Report. (Yubunsha Co., Ltd., 1-5, Kanda
Suda-Cho, Chiyoda-ku, KS Bldg., Tokyo 101, Japan) V.1- 1960-
Volume(issue)/page/year: 22,1419,1988
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 1650 mg/kg
- SEX/DURATION :
- female 7-17 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Fertility - post-implantation mortality (e.g. dead and/or
resorbed implants per total number of implants)
Reproductive - Fertility - litter size (e.g. # fetuses per litter; measured
before birth)
Reproductive - Effects on Embryo or Fetus - fetal death
- REFERENCE :
- KSRNAM Kiso to Rinsho. Clinical Report. (Yubunsha Co., Ltd., 1-5, Kanda
Suda-Cho, Chiyoda-ku, KS Bldg., Tokyo 101, Japan) V.1- 1960-
Volume(issue)/page/year: 13,1160,1979
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravenous
- DOSE :
- 13 mg/kg
- SEX/DURATION :
- female 6-18 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Fertility - pre-implantation mortality (e.g. reduction in
number of implants per female; total number of implants per corpora lutea)
Reproductive - Effects on Embryo or Fetus - fetal death
- REFERENCE :
- KSRNAM Kiso to Rinsho. Clinical Report. (Yubunsha Co., Ltd., 1-5, Kanda
Suda-Cho, Chiyoda-ku, KS Bldg., Tokyo 101, Japan) V.1- 1960-
Volume(issue)/page/year: 20,4641,1986